Researchers Revel in C9ORF72 Advances at RNA Symposium
Quick Links
Researchers interested in amyotrophic lateral sclerosis and other disorders of RNA arrived early for the Society for Neuroscience annual meeting this year, attending “RNA Metabolism in Neurological Disease,” a two-day satellite symposium held November 7-8 in San Diego. To facilitate cross-fertilization among researchers studying different kinds of repeat disorders and RNA metabolism pathology, meeting organizers Paul Taylor of St. Jude Children’s Research Hospital in Memphis, Tennessee, and Fen-Biao Gao of the University of Massachusetts Medical School in Worcester invited speakers studying a range of conditions. Gao told Alzforum he was glad to see attendance up from the last RNA meeting in 2011 (see Nov 2011 conference story), with nearly 300 attendees.
About one-third of the more than 30 genes linked to ALS affect RNA metabolism, either directly or indirectly, said Robert Brown of the University of Massachusetts Medical School in Worcester, who opened the conference with an overview of genetics. Many attendees were excited about recent results in the study of the C9ORF72 gene. C9ORF72 hexanucleotide repeat expansions are the most common cause of familial ALS and frontotemporal dementia. Scientists discussed how the repeat size correlates with disease presentation, various up-and-coming animal models, and potential treatment strategies. Antisense oligonucleotides were recently tested in disease models as one possible treatment strategy (see Oct 2013 news story; news story). Christopher Pearson of The Hospital for Sick Children in Toronto suggested another, based on destabilizing G-quadruplex structures that the repeat likely assumes.
Pearson and others noted that ALS and FTD researchers benefit from previous studies of more than 40 other nucleotide repeat disorders, such as myotonic dystrophy, from which they can mine hypotheses and techniques. For example, repeat associated non-ATG (RAN) translation, identified for C9ORF72 earlier this year, was found first in other diseases (see Feb 2013 news story, news story). Laura Ranum of the University of Florida in Gainesville, who originally described this unusual translation, welcomed C9ORF72 researchers to the fold (Zu et al., 2011). “The RAN translation field has been pretty lonely,” she said.
A Stable Shape for C9ORF72 RNA Foci?
Scientists have established that C9ORF72 forms stable RNA foci, with aggregates forming out of RNA in both the sense and antisense direction (see Nov 2013 news story). How might the GGGGCC repeats tangle together? Pearson presented results, released over the last year by his group and others, that suggest the sense RNA forms a stable structure called a G-quadruplex (see Sep 2012 news story, Reddy et al., 2013, Fratta et al., 2013).

Stabilizing Expansions
G-quadruplexes are formed of stacks of square G-quartets, with guanines at each corner, surrounding a positive ion. The DNA or RNA strands forming the edges can be parallel or antiparallel, as shown here, and come from the same nucleic acid chain (as shown) or multiple chains. Nucleotides that are not guanines hang off the structure in the external loops. [Image courtesy of Julian Huppert, Wikimedia Commons.]
G-quadruplexes form naturally from guanine-rich sequences of single-stranded DNA or RNA. Four guanines interact around a positive ion, making a square called a G-quartet, and these stacked squares make up a quadruplex. An individual nucleic acid strand may form a quadruplex, or multiple strands may come together to create it. G-quadruplexes appear throughout biology. For example, unimolecular G-quadruplexes show up in the single-stranded telomeric DNA at the ends of chromosomes, and in oncogene promoters when they unwind DNA (reviewed in Chen and Yang, 2012). In RNA, G-quadruplexes are common in 5’ untranslated sequences and affect translation (reviewed in Bugaut and Balasubramanian, 2012).
Given the guanine-rich nature of the C9ORF72 repeat sequence, Pearson suspected it might form a G-quadruplex. Using circular dichroism, Kaalak Reddy and colleagues in Pearson's lab showed this was the case for repeats of two to eight GGGGCC sequences in RNA in vitro. The structures formed out of multiple RNAs, with the backbones of the RNA at each corner of the quadruplex lined up in parallel. The G-quadruplexes were highly stable, and did not melt completely even at 95 degrees Celsius.
Researchers suspect that C9ORF72 expanded RNA forms these structures in vivo, though it would be difficult to prove, Pearson told Alzforum. He also found that some RNA-binding proteins, such as hnRNPA1, interact with C9ORF72 quadruplexes in vitro. The antisense version of the expanded RNA (GGCCCC), which researchers recently found transcribed in cells expressing expanded C9ORF72, would be more likely to form a hairpin, he predicted.
If RNA foci comprising G-quadruplexes prove to be toxic, then drugs that bind the quadruplex backbone might do some good, Pearson reasoned. Researchers have already developed some small molecules that interact with the quadruplexes (reviewed in Neidle, 2010). Pearson and colleagues tested one such compound on C9ORF72 G-quadruplexes in vitro (see Siddiqui-Jain et al., 2002; Monchaud et al., 2010; Faudale et al., 2012). TMPyP4—which stands for the mouthful tri-meso(N-methyl-4-pyridyl), meso(N-tetradecyl-4-pyridyl) porphine—looks like a four-pronged “ninja star,” Pearson said, that can stick to the squares of G-quadruplexes. He found it bound C9ORF72 G-quadruplexes and destabilized them. It also interrupted the interaction between the quadruplex and RNA-binding proteins such as hnRNPA1, he reported.
Brown said Pearson’s ideas should be tested in cells and animals as soon as possible. However, Pearson was careful to caution that he does not propose TMPyP4 as a treatment for human disease, but rather presents the results as a proof of principle that a small molecule can alter the structure of C9ORF72 RNA foci. For one, he pointed out, researchers are not yet certain if RNA foci are the pathological cause of C9ORF72 disease. For another, TMPyP4 binds several kinds of G-quadruplexes, not just the GGGGCC sequence, and he does not yet know if it binds C9ORF72’s sequence preferentially.
C9ORF72 Genetics
While Pearson worked with a handful of repeats in vitro, in people the expansion can grow to considerable lengths. Marka van Blitterswijk and colleagues in Rosa Rademakers' lab at the Mayo Clinic in Jacksonville, Florida, have characterized the repeat size in different tissues of individuals who carry the expansion. As Rademakers’ lab reported previously, the number of repeats varies widely among expansion carriers and even among tissues within the same person (see Jan 2013 conference story). Van Blitterswijk and colleagues used Southern blots to measure repeat sizes in 84 expansion carriers with FTD, motor neuron disease, or both, as well as some asymptomatic carriers, from a range of tissues including blood, cerebellum, and frontal cortex, as she presented in a poster and Rademakers showed in a slide presentation (see Van Blitterswijk et al., 2013). Overall, the cerebellum tended to have the shortest average repeat length, 1,667, compared with 5,250 in frontal cortex. The expansion size failed to predict whether someone had FTD versus ALS. Blood repeat size, with an average of 2,717, correlated with neither brain repeat length nor disease severity. Pearson said this was disappointing from a clinical point of view, because it precludes predicting disease course with a simple blood test for expansion size. However, blood testing can still identify or exclude someone as an expansion carrier, Van Blitterswijk noted.
Van Blitterswijk and others have reported that C9ORF72 expansion carriers may have second mutations that could contribute to pathology, and perhaps even explain why one carrier gets FTD while another has ALS (see Jan 2013 conference story; Van Blitterswijk et al., 2012; Van Blitterswijk et al., 2013). In San Diego, Rademakers reported genotyping C9ORF72 carriers for a rare variant in the gene TMEM106B, a protective factor against FTD in carriers of progranulin mutations (see Feb 2010 news story; Finch et al., 2011; Aug 2012 news story). Among more than 300 expansion carriers, Van Blitterswijk observed a lower-than-normal frequency of homozygotes for the minor allele, but this pattern occurred only in those with FTD. The protective, minor allele appears to stave off FTD, but not ALS, she suggested.
TMEM106B is the first genetic modifier of C9ORF72 to be identified, Rademakers noted. Brown agreed. “It looks like it is a real modifier,” he said. Van Blitterswijk speculated that because TMEM106B influences lysosome shape and activity, genetic variants might affect degradation and aggregation of TDP-43, which could influence FTD risk.
Animal Models Under Construction
Since the discovery of the C9ORF72 gene’s link to disease in 2011 (see Sep 2011 news story), researchers have started to develop a number of experimental models to study the expansion. Gao, Christopher Donnelly of Johns Hopkins University in Baltimore, and Clotilde Lagier-Tourenne of the University of California, San Diego, described published models of cultured primary fibroblast and induced pluripotent stem cell lines from C9ORF72 expansion carriers (see Almeida et al., 2013). In San Diego, researchers reported progress with several new whole-animal models. The creatures are difficult to create, scientists told Alzforum, due to the unstable nature of the repeated sequence.
Brian Freibaum of St. Jude and Helene Tran of the University of Massachusetts in Worcester reported introducing C9ORF72 into fruit flies, which have no homolog to the human gene. Freibaum expressed C9ORF72 with eight, 28, and 58 repeats (the cutoff between health and disease appears to be about 30) and looked for RNA aggregates and peptides due to translation of the repeat sequence, both of which are subjects of intensive research. At eight repeats, he saw neither. At 28, he observed RNA foci. At 58, he saw both RNA foci and peptide aggregates. The 28-repeat construct might be “on the cusp” of RAN translation, Freibaum speculated. The longer constructs were toxic when expressed in various fly tissues. For example, flies transcribing C9ORF72 with 28 or 58 repeats in muscle tissue held their wings awkwardly up or down, instead of flat like wild-type animals.

Peptides RAN amok in flies
A proline-glycine poly-dipeptide (green) born of RAN translation of C9ORF72 aggregates in the muscles of transgenic Drosophila. Image courtesy of Brian Freibaum, St. Jude Children’s Research Hospital, Memphis, Tennessee.
Tran engineered flies with a range of repeat lengths. Those with 160 repeats accumulated RNA aggregates, climbed poorly, and died earlier than flies with only five repeats. Both Tran and Freibaum are conducting screens for genetic suppressors and enhancers of the C9ORF72 phenotype in their models. These screens could identify genes involved in disease progression and potential drug targets, Freibaum told Alzforum. The repeats also caused neurodegeneration in a fly model published earlier this year (see Xu et al., 2013).
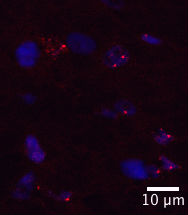
Focus on Flies
In a Drosophila model of C9ORF72 with 160 repeats, fluorescence in-situ hybridization identifies RNA foci (red) in the ventral nerve cord of larvae (blue=nuclei). Image courtesy of Helene Tran, University of Massachusetts, Worcester.
Vertebrate C9ORF72 models are also making headway. Bettina Schmid of the German Center for Neurodegenerative Diseases (DZNE) in Munich presented preliminary results from zebrafish models. Schmid created knockout animals and fish that express two or 80 repeats in the zebrafish homolog of the human gene. Knockout models should help identify the natural function of C9ORF72, which is unknown, Schmid told Alzforum. However, her knockouts had no obvious phenotype. “They swim and grow normally,” Schmid said. Another group recently reported that zebrafish C9ORF72 knockouts swam poorly. The difference could be due to different knockout technology used by the two groups, Schmid speculated (see Ciura et al., 2013). In Schmid’s expansion models, two repeats elicited no RNA foci or peptide aggregates, whereas 80 repeats created both. Schmid says she needs to confirm these preliminary results.
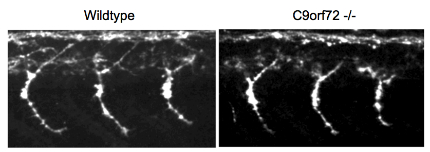
No C9ORF72, No Problem
Motor neuron axon length was the same between wild-type and C9ORF72 knockout zebrafish embryos in these sections stained with an antibody to znp-1. Image courtesy of Alexander Hruscha, German Center for Neurodegenerative Diseases, Munich.
Finally, both Brown and Lagier-Tourenne discussed mouse models. They and several other researchers are part of a large collaboration, working with C9ORF72 bacterial artificial chromosomes provided by the ALS Association. Brown described a mouse engineered to have 600 repeats which has now reached 11 months old and produced three further generations. It has no obvious motor phenotype, he reported. The animals gain weight normally and grip and balance on a rotating rod just like wild-type mice. However, Brown believes the animals will be useful because they accumulate RNA foci and RAN-translated dipeptides, as collaborators in Gao’s lab and Leonard Petrucelli's lab at the Mayo Clinic in Jacksonville have found. Lagier-Tourenne and Don Cleveland at the University of California, San Diego, have created several mouse lines, with repeats ranging from 100 to 450. They plan to test antisense oligonucleotide treatments in those mice.
Brown also reported on a C9ORF72 knockout model created by Kevin Eggan of Harvard University. These mice are about a year old, and the researchers have observed a subtle but progressive loss of motor function, including denervation of muscles, paralysis, and early death. These data support the theory that C9ORF72 disease results from haploinsufficiency of the gene, commented Aaron Gitler of Stanford University in Palo Alto, California. Lucie Bruijn from the ALS Association said that all these models would be useful and complementary.
Future Questions
Researchers at this meeting agreed that they have only started to untangle the effects of C9ORF72 expansions. “Much more needs to be understood at the mechanistic level,” Gao told Alzforum. “We know so little.” Some data support loss of C9ORF72 function as a problem, but what about RNA foci and RAN-translated peptides—“Are they pathogenic, or are they a parlor trick?” asked Brown. He said for now data seem to indicate that silencing C9ORF72 would be beneficial, and Bruijn agreed this would be her priority treatment approach.—Amber Dance
References
News Citations
- DC: New ALS Genetics Hog the Limelight at Satellite Conference
- RNA Deposits Confer Toxicity in C9ORF72 ALS
- Second Study Confirms Antisense Oligonucleotides Bust RNA Aggregates
- RNA Twist: C9ORF72 Intron Expansion Makes Aggregating Protein
- Second Study Sees Intron in FTLD Gene Translated
- Sense, Antisense: C9ORF72 Makes Both Forms of RNA, Peptides
- C9ORF72 Steals the Show at Frontotemporal Dementia Meeting
- Chicago—Dynamic Repeats: C9ORF72 Expands and Shrinks in ALS
- Chicago—Devilish Duo: Two Mutations Add Up to Familial ALS
- Genetics of FTD: New Gene, PGRN Variety, and a Bit of FUS
- FTD Risk Factor Confirmed, Alters Progranulin Pathways
- Corrupt Code: DNA Repeats Are Common Cause for ALS and FTD
Paper Citations
- Zu T, Gibbens B, Doty NS, Gomes-Pereira M, Huguet A, Stone MD, Margolis J, Peterson M, Markowski TW, Ingram MA, Nan Z, Forster C, Low WC, Schoser B, Somia NV, Clark HB, Schmechel S, Bitterman PB, Gourdon G, Swanson MS, Moseley M, Ranum LP. Non-ATG-initiated translation directed by microsatellite expansions. Proc Natl Acad Sci U S A. 2011 Jan 4;108(1):260-5. Epub 2010 Dec 20 PubMed.
- Reddy K, Zamiri B, Stanley SY, Macgregor RB, Pearson CE. The disease-associated r(GGGGCC)n repeat from the C9orf72 gene forms tract length-dependent uni- and multimolecular RNA G-quadruplex structures. J Biol Chem. 2013 Apr 5;288(14):9860-6. PubMed.
- Fratta P, Mizielinska S, Nicoll AJ, Zloh M, Fisher EM, Parkinson G, Isaacs AM. C9orf72 hexanucleotide repeat associated with amyotrophic lateral sclerosis and frontotemporal dementia forms RNA G-quadruplexes. Sci Rep. 2012;2:1016. PubMed.
- Chen Y, Yang D. Sequence, stability, and structure of G-quadruplexes and their interactions with drugs. Curr Protoc Nucleic Acid Chem. 2012 Sep;Chapter 17:Unit17.5. PubMed.
- Bugaut A, Balasubramanian S. 5'-UTR RNA G-quadruplexes: translation regulation and targeting. Nucleic Acids Res. 2012 Jun;40(11):4727-41. Epub 2012 Feb 20 PubMed.
- Neidle S. Human telomeric G-quadruplex: the current status of telomeric G-quadruplexes as therapeutic targets in human cancer. FEBS J. 2010 Mar;277(5):1118-25. PubMed.
- Siddiqui-Jain A, Grand CL, Bearss DJ, Hurley LH. Direct evidence for a G-quadruplex in a promoter region and its targeting with a small molecule to repress c-MYC transcription. Proc Natl Acad Sci U S A. 2002 Sep 3;99(18):11593-8. PubMed.
- Monchaud D, Granzhan A, Saettel N, Guédin A, Mergny JL, Teulade-Fichou MP. "One ring to bind them all"-part I: the efficiency of the macrocyclic scaffold for g-quadruplex DNA recognition. J Nucleic Acids. 2010;2010 PubMed.
- Faudale M, Cogoi S, Xodo LE. Photoactivated cationic alkyl-substituted porphyrin binding to g4-RNA in the 5'-UTR of KRAS oncogene represses translation. Chem Commun (Camb). 2012 Jan 21;48(6):874-6. PubMed.
- van Blitterswijk M, DeJesus-Hernandez M, Niemantsverdriet E, Murray ME, Heckman MG, Diehl NN, Brown PH, Baker MC, Finch NA, Bauer PO, Serrano G, Beach TG, Josephs KA, Knopman DS, Petersen RC, Boeve BF, Graff-Radford NR, Boylan KB, Petrucelli L, Dickson DW, Rademakers R. Association between repeat sizes and clinical and pathological characteristics in carriers of C9ORF72 repeat expansions (Xpansize-72): a cross-sectional cohort study. Lancet Neurol. 2013 Oct;12(10):978-88. Epub 2013 Sep 5 PubMed.
- van Blitterswijk M, van Es MA, Hennekam EA, Dooijes D, van Rheenen W, Medic J, Bourque PR, Schelhaas HJ, van der Kooi AJ, de Visser M, de Bakker PI, Veldink JH, van den Berg LH. Evidence for an oligogenic basis of amyotrophic lateral sclerosis. Hum Mol Genet. 2012 Sep 1;21(17):3776-84. Epub 2012 May 29 PubMed.
- van Blitterswijk M, Baker MC, Dejesus-Hernandez M, Ghidoni R, Benussi L, Finger E, Hsiung GY, Kelley BJ, Murray ME, Rutherford NJ, Brown PE, Ravenscroft T, Mullen B, Ash PE, Bieniek KF, Hatanpaa KJ, Karydas A, Wood EM, Coppola G, Bigio EH, Lippa C, Strong MJ, Beach TG, Knopman DS, Huey ED, Mesulam M, Bird T, White CL, Kertesz A, Geschwind DH, Van Deerlin VM, Petersen RC, Binetti G, Miller BL, Petrucelli L, Wszolek ZK, Boylan KB, Graff-Radford NR, Mackenzie IR, Boeve BF, Dickson DW, Rademakers R. C9ORF72 repeat expansions in cases with previously identified pathogenic mutations. Neurology. 2013 Oct 8;81(15):1332-41. PubMed.
- Finch N, Carrasquillo MM, Baker M, Rutherford NJ, Coppola G, Dejesus-Hernandez M, Crook R, Hunter T, Ghidoni R, Benussi L, Crook J, Finger E, Hantanpaa KJ, Karydas AM, Sengdy P, Gonzalez J, Seeley WW, Johnson N, Beach TG, Mesulam M, Forloni G, Kertesz A, Knopman DS, Uitti R, White CL, Caselli R, Lippa C, Bigio EH, Wszolek ZK, Binetti G, Mackenzie IR, Miller BL, Boeve BF, Younkin SG, Dickson DW, Petersen RC, Graff-Radford NR, Geschwind DH, Rademakers R. TMEM106B regulates progranulin levels and the penetrance of FTLD in GRN mutation carriers. Neurology. 2011 Feb 1;76(5):467-74. PubMed.
- Almeida S, Gascon E, Tran H, Chou HJ, Gendron TF, Degroot S, Tapper AR, Sellier C, Charlet-Berguerand N, Karydas A, Seeley WW, Boxer AL, Petrucelli L, Miller BL, Gao FB. Modeling key pathological features of frontotemporal dementia with C9ORF72 repeat expansion in iPSC-derived human neurons. Acta Neuropathol. 2013 Sep;126(3):385-99. PubMed.
- Xu Z, Poidevin M, Li X, Li Y, Shu L, Nelson DL, Li H, Hales CM, Gearing M, Wingo TS, Jin P. Expanded GGGGCC repeat RNA associated with amyotrophic lateral sclerosis and frontotemporal dementia causes neurodegeneration. Proc Natl Acad Sci U S A. 2013 May 7;110(19):7778-83. PubMed.
- Ciura S, Lattante S, Le Ber I, Latouche M, Tostivint H, Brice A, Kabashi E. Loss of function of C9orf72 causes motor deficits in a zebrafish model of Amyotrophic Lateral Sclerosis. Ann Neurol. 2013 May 30; PubMed.
Further Reading
Papers
- Suzuki N, Maroof AM, Merkle FT, Koszka K, Intoh A, Armstrong I, Moccia R, Davis-Dusenbery BN, Eggan K. The mouse C9ORF72 ortholog is enriched in neurons known to degenerate in ALS and FTD. Nat Neurosci. 2013 Dec;16(12):1725-7. Epub 2013 Nov 3 PubMed.
- Galimberti D, Arosio B, Fenoglio C, Serpente M, Cioffi SM, Bonsi R, Rossi P, Abbate C, Mari D, Scarpini E. Incomplete Penetrance of the C9ORF72 Hexanucleotide Repeat Expansions: Frequency in a Cohort of Geriatric Non-Demented Subjects. J Alzheimers Dis. 2013 Oct 10; PubMed.
- Dols-Icardo O, García-Redondo A, Rojas-García R, Sánchez-Valle R, Noguera A, Gómez-Tortosa E, Pastor P, Hernández I, Esteban-Pérez J, Suárez-Calvet M, Antón-Aguirre S, Amer G, Ortega-Cubero S, Blesa R, Fortea J, Alcolea D, Capdevila A, Antonell A, Lladó A, Muñoz-Blanco JL, Mora JS, Galán-Dávila L, Rodríguez de Rivera FJ, Lleó A, Clarimón J. Characterization of the repeat expansion size in C9orf72 in amyotrophic lateral sclerosis and frontotemporal dementia. Hum Mol Genet. 2013 Oct 8; PubMed.
- Mackenzie IR, Arzberger T, Kremmer E, Troost D, Lorenzl S, Mori K, Weng SM, Haass C, Kretzschmar HA, Edbauer D, Neumann M. Dipeptide repeat protein pathology in C9ORF72 mutation cases: clinico-pathological correlations. Acta Neuropathol. 2013 Dec;126(6):859-79. Epub 2013 Oct 6 PubMed.
- Ticozzi N, Tiloca C, Calini D, Gagliardi S, Altieri A, Colombrita C, Cereda C, Ratti A, Pezzoli G, Borroni B, Goldwurm S, Padovani A, Silani V. C9orf72 repeat expansions are restricted to the ALS-FTD spectrum. Neurobiol Aging. 2014 Apr;35(4):936.e13-7. Epub 2013 Oct 2 PubMed.
News
- Brain Imaging Distinguishes C9ORF72 From Other Types of ALS
- Methylation a Turn Off for Disease Gene C9ORF72?
- RNA Twist: C9ORF72 Intron Expansion Makes Aggregating Protein
- C9ORF72 Function: Is the ALS Protein a Membrane Traffic Cop?
- Chicago—RNA Inclusions Offer Therapeutic Target in ALS
- When Is a C9ORF72 Repeat Expansion Not a C9ORF72 Repeat Expansion?
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.