Longitudinal Data Stand Out at AD/PD Imaging Satellite
Quick Links
Read a PDF of the entire series.
This is Part 1 of a two-part series. See also Part 2.
In a sign of how brain imaging is increasingly infiltrating clinical research on age-related neurodegeneration, the 11th AD/PD International Conference on Alzheimer’s and Parkinson’s Diseases hosted a day-long exchange of new imaging data and discussion of current challenges for the research community in these two disorders. On 5 March 2013, in a renovated space inside the Fortezza da Basso—called “Basilica” for its arched Roman columns, central nave and wings—several hundred researchers met to update each other on the latest in their field. Co-organized by Mike Weiner of the University of California, San Francisco, and Ken Marek, at the Institute for Neurodegenerative Disorders in New Haven, Connecticut, the day consolidated an emerging consensus that in both diseases, outwardly normal people with preclinical imaging evidence of AD or PD face an elevated risk of developing full-fledged disease. This enables researchers to stage people early on. Consequently, a growing number of clinical trials is taking advantage of brain imaging to better define whom to enroll. In contrast, trial designers are struggling with how to use imaging to measure a drug’s effect. More tracers are coming online and are being used on related diseases such as Down’s syndrome. Meanwhile, in the day-to-day work of memory clinics, imaging appears poised—for better or worse, and at considerable cost—to gradually edge out clinical/neuropsychological evaluation in the years to come. “The clinical diagnosis is often wrong. It may become secondary to imaging,” Weiner said. Here are selected highlights.
Chris Rowe of Melbourne University, Australia, shared the latest imaging results from the Australian Imaging, Biomarkers & Lifestyle Study of Ageing. One of the largest longitudinal brain imaging studies around the globe, AIBL engages some 100 scientists and, like ADNI, has brought academia and industry together. Starting in 2006, AIBL researchers ran serial PIB-PET and MRI scans on 288 participants; they have recently increased this number to 750 people.
Prospective clinical studies elsewhere are adding on imaging as well; for example, the Mayo Clinic Study of Aging now runs scans on some 500 of its 2,700 participants. Similar studies include the longstanding Washington University Memory & Aging Project, the Harvard Aging Brain Study, Wisconsin Registry for Alzheimer’s Prevention, Parkinson’s Progression Markers Initiative, as well as De Novo Parkinson, DESCRIPA, and Innovative Medicines Initiative (IMI). They teach researchers how AD and PD develop. For AD, they are testing and refining a popular staging model that is based on hypothetical and cross-sectional data (see ARF related news story).
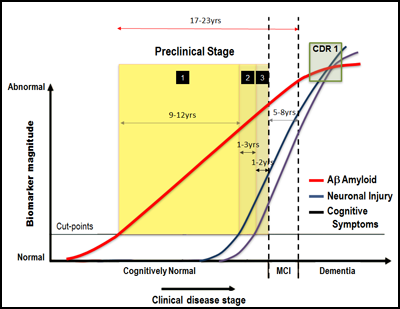
In Florence, Rowe presented the three- to six-year follow-up from AIBL, the largest batch of longitudinal data to date. He showed how some people accumulate brain amyloid quickly, others slowly, and a third group not at all. The Australian researchers calculated how fast this deposition grows, and how it relates to other markers and symptoms of AD. This work appeared in Lancet Neurology during AD/PD 2013 and was widely discussed at the conference (see ARF companion story). In essence, it takes the average "accumulator" some 12 years to go from a negative scan to the threshold of amyloid positivity, and 19 more years from there to Alzheimer’s dementia. Brain atrophy and memory change much later in the process. When applied to the proposed NIA-AA diagnostic criteria and expressed in their terminology (Sperling et al., 2011; Albert et al., 2011), these data generate a staging diagram with a drawn-out "preclinical" stage of 11-17 years and a five- to eight-year "MCI due to AD" stage. In short, longitudinal AIBL data support new diagnostic criteria that no longer require dementia to make a diagnosis of Alzheimer’s disease, Rowe said.
When fit to new diagnostic criteria, AIBL suggests a preclinical stage of 11-17 years and a mildly symptomatic phase of five to eight years. Image courtesy of AIBL
How can these types of data help doctors formulate a prognosis for individual patients? The field is not there yet, but AIBL is contributing pieces, Rowe said. Following people over time shows that cognitively normal people with brain amyloid decline on repeat testing, whereas amyloid-negative people improve with practice. “Our data support the concept of prodromal or very early AD. People with brain amyloid tend to have poor episodic memory,” said Rowe.
Amyloid positivity is not a simple, binary thing, even though that is largely how such scans may be read in routine clinical practice. In AIBL, the amount of amyloid people had at baseline strongly influenced their likelihood of progressing. Having a lot is more worrisome than having a little. “We think it is important how much amyloid is present, not just whether you are positive or negative on your scan. Being strongly positive is much worse than just being positive,” said Rowe. This finding argues against attaching dire predictions to a barely positive scan. This is one reason why clinical practice guidelines issued this past January argue against offering predictive amyloid scans to cognitively normal people at this time (see ARF related news story).
Because the initial amyloid burden at first scan predicts a person’s progression more strongly than the rate at which it grows, there is no point in serial PET scans for diagnostic purposes. An amyloid scan costs several thousand dollars, and for clinical purposes a second scan generally won’t offer more informative data than the first, Rowe said. Serial MRI scans are a common practice.
Repeat scanning to see how fast people’s amyloid load is growing poorly predicts whether they will be become symptomatic; more can be learned from a single scan measuring exactly how much amyloid they already have. Image courtesy of AIBL
Rowe focused on how amyloid deposition relates to cognitive performance over time. At baseline, differences among the cognitively normal volunteers were difficult to pin down even though the amyloid-positive subgroup did do slightly worse, Rowe said; however, over the next few years, the amyloid-positive people clearly declined. He presented data showing that a cognitively normal people’s odds of meeting diagnostic criteria for MCI due to AD within three years go up 4.8-fold if they have brain amyloid; the odds of progressing from MCI to AD dementia shoot up 14-fold.
Brain amyloid is not harmless. Image courtesy of AIBL
AIBL data are being used to figure out which combination of markers most reliably predicts a person’s likely course over the next few years. Other longitudinal studies are doing this as well. Much of this work currently focuses on people who already are cognitively impaired but still function normally, partly because this MCI stage is where most data exist, and partly because clinical trials are increasingly targeting this population. With regard to who among the MCI cases is on the path to Alzheimer’s dementia, a combination of abnormal markers tends to be more informative than a single marker.
In old story: In Florence, Rowe showed AIBL data suggesting that, for participants who had MCI at baseline, having hippocampal atrophy or carrying ApoE4 ups their odds of dementia within three years by a factor of 4 and 5, respectively. Being impaired on verbal learning and memory boosts that odds ratio to 11, being positive on amyloid PET to 15-fold. The combination of brain amyloid and hippocampal atrophy boosts the odds of developing dementia 44-fold. If a person had brain amyloid and was amnestic, then dementia was virtually guaranteed, with an odds ratio of more than a million, Rowe said.
Other studies are largely convergent. The up to six-year follow-up of the European DESCRIPA collaborative found that its 900 MCI patients seen at 20 clinics across the continent comprised—as did AIBL's—non-progressors and progressors. People who at baseline were abnormal on all three biomarkers measured—in this case amyloid pathology via CSF, brain activity via FDG-PET, and hippocampal shrinkage via MRI—were certain of declining. In essence, they already had AD, just not yet its dementia (partially published Vos et al., 2013; Prestia et al., 2013). ADNI data led to much the same conclusion in a recent analysis of 97 participants. The presence of β amyloid added the single biggest predictive value, but total certainty came from the combination of amyloid, MRI, and an episodic memory deficit, said Murali Doraiswamy of Duke University in Durham, North Carolina (Shaffer et al., 2013).
In Florence, these data sparked discussion about whether clinicians should stop first doing structural MRI scans in people who they suspect have MCI. Should they go straight for an amyloid PET scan? After all, MRI usually requires serial scans to aid prognosis. Some research physicians felt the current practice of MRI first was still appropriate because it can show vascular pathology or other lesions as the reason for the patient’s symptoms. Others suspected that structural MRI for MCI might be on its way out, because many PET scanners in clinical settings can spot tumors and strokes or other lesions, as well.
To Rowe, the convergent progression data mean that MCI is a symptom, not a diagnosis. “But cognitive impairment is what the clinician is faced with, so it is a reasonable group to recruit and follow. With biomarkers, we can pull its heterogeneous causes apart and make real diagnoses,” Rowe said.
Like many groups around the world, AIBL, too, sees a percentage of people with a clinical phenotype of MCI or mild AD whose scan is negative for β amyloid. What do these people have? Their outcomes after three to five years have been dementia with Lewy bodies, the tauopathy supranuclear palsy, frontotemporal dementia, and vascular dementia. Some participants never fit an alternative diagnosis, said Rowe. Some reverted to normal, while others declined until they had to enter a nursing home with an unspecified dementia. Other research groups are finding the same thing, said Doraiswamy, who considers a negative amyloid scan in people with symptoms of MCI particularly useful in the clinic. To parse MCI more completely and predict what will become of the non-AD cases, scientists need tracers for α-synuclein, tau, or the requisite neurotransmitters.
The symposium, as well as the AD/PD conference itself, featured repeated calls for more molecular diagnostics to help clinicians diagnose neurodegenerative diseases more precisely, and to help researchers understand how various pathogenic proteins interact to cause the AD-PD spectrum of illnesses. Postmortem pathology for years has reminded doctors that, at least by the time patients die, many cases of dementia are mixed. Newly issued pathology guidelines have reinforced this message, said Dennis Dickson at the Mayo Clinic in Jacksonville, Florida.
Research on tau tracers is diversifying, with compounds from Japan’s Tohoku University, that country’s Molecular Imaging Center in Chiba, and Siemens Molecular Imaging in early stages of human testing. In addition, Merck is in the middle of a “big effort to screen 3.5 million compounds,” said Richard Hargreaves of that company. On α-synuclein, a Michael J Fox Foundation-funded consortium between researchers at the contract research organization BioFocus near Cambridge, U.K., and U.S. experts including Robert Mach, Bill Klunk, and Chet Mathis has made headway with lead compounds that bind aggregated α-synuclein fibrils; none of them is in human testing yet.
Parkinson’s disease researchers have a molecular diagnostic tool in β-CIT, aka DAT scan. It quantifies the PD-related shrinkage of dopamine transporters in the putamen and striatum areas of the brain. It has been used in trials; however, like amyloid PET tracers, it changes slowly, declining at a rate of about 4 percent per year. “We need imaging markers that show clear change within six months,” said Marek. DAT scans are helping scientists dissect the reasons why people with Parkinson’s get dementia later on (see ARF related news story), and they are helping researchers enrich populations for early-stage trials at the newly redefined premotor and preclinical stages (Stern et al., 2012). Finding such patients is arduous in PD because its incidence is lower than that of AD, said Andrew Siderowf, who recently left the University of Pennsylvania to work for Avid Radiopharmaceuticals, both in Philadelphia.
In Florence, numerous researchers applauded natural history studies such as AIBL. “We should take advantage of the enormous insight we get from longitudinal cohorts, and maintain them as best we can,” said Marek, who leads the PPMI. AIBL is enrolling new people as its advanced patients drop out or die, keeping the study at 1,000 participants; ADNI is currently recruiting for its second phase (see ARF ADNI update story). Industry likes those studies, too, as is evident from their funding of ADNI, AIBL, and PPMI. “We need to follow many subjects examined with biomarkers to autopsy,” said Merck’s Hargreaves.—Gabrielle Strobel.
This is Part 1 of a two-part series. See also Part 2.
References
News Citations
- Brain Imaging in Trials—How to Make It Work?
- HAI—Sharper Curves: Revamping a Biomarker Staging Model
- From Natural History, A "Renaissance" for Amyloid Hypothesis
- HAI—Amyloid Imaging in the Clinic: New Guidelines and Data
- Dementia in Movement Disorders: What Causes It?
- Wrapping Up Enrollment, ADNI 2, PPMI Focus on Earlier Disease
Paper Citations
- Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, Iwatsubo T, Jack CR Jr, Kaye J, Montine TJ, Park DC, Reiman EM, Rowe CC, Siemers E, Stern Y, Yaffe K, Carrillo MC, Thies B, Morrison-Bogorad M, Wagster MV, Phelps CH. Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011 May;7(3):280-92. Epub 2011 Apr 21 PubMed.
- Albert MS, DeKosky ST, Dickson D, Dubois B, Feldman HH, Fox NC, Gamst A, Holtzman DM, Jagust WJ, Petersen RC, Snyder PJ, Carrillo MC, Thies B, Phelps CH. The diagnosis of mild cognitive impairment due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011 May;7(3):270-9. Epub 2011 Apr 21 PubMed.
- Vos SJ, van Rossum IA, Verhey F, Knol DL, Soininen H, Wahlund LO, Hampel H, Tsolaki M, Minthon L, Frisoni GB, Froelich L, Nobili F, van der Flier W, Blennow K, Wolz R, Scheltens P, Visser PJ. Prediction of Alzheimer disease in subjects with amnestic and nonamnestic MCI. Neurology. 2013 Mar 19;80(12):1124-32. PubMed.
- Prestia A, Caroli A, van der Flier WM, Ossenkoppele R, Van Berckel B, Barkhof F, Teunissen CE, Wall AE, Carter SF, Schöll M, Choo IH, Nordberg A, Scheltens P, Frisoni GB. Prediction of dementia in MCI patients based on core diagnostic markers for Alzheimer disease. Neurology. 2013 Mar 12;80(11):1048-56. Epub 2013 Feb 6 PubMed.
- Shaffer JL, Petrella JR, Sheldon FC, Choudhury KR, Calhoun VD, Coleman RE, Doraiswamy PM, . Predicting cognitive decline in subjects at risk for Alzheimer disease by using combined cerebrospinal fluid, MR imaging, and PET biomarkers. Radiology. 2013 Feb;266(2):583-91. PubMed.
- Stern MB, Lang A, Poewe W. Toward a redefinition of Parkinson's disease. Mov Disord. 2012 Jan;27(1):54-60. PubMed.
Other Citations
External Citations
Further Reading
News
- Tau, α-Synuclein Spread: Crazy Stuff—How Might It Work?
- Like Star Born of Supernova, Plaque Born of Exploded Neuron?
- Can Dousing PyroGlu-Aβ Treat Alzheimer’s Disease?
- Field Ramps Up "Mini" Mouse MRI
- LRRK Watchers’ Eyes Turn to Inflammation, Autophagy, Kinase
- Can Cancer Therapy Be Neurodegenerative Wonder Drug?
- Taking Aim at M1: Old Hat or New Target?
- Dementia in Movement Disorders: What Causes It?
- BACE Inhibitors Barrel Forward—Next Hurdles: Safety, Efficacy
- In Pursuit of Toxic Tau
- Sleep Patterns, Circadian Clock Linked to Aβ Oxidative Stress
- HAI—Sharper Curves: Revamping a Biomarker Staging Model
- From Natural History, A "Renaissance" for Amyloid Hypothesis
- Wrapping Up Enrollment, ADNI 2, PPMI Focus on Earlier Disease
- HAI—Amyloid Imaging in the Clinic: New Guidelines and Data
Annotate
To make an annotation you must Login or Register.



Comments
No Available Comments
Make a Comment
To make a comment you must login or register.