Tunneling Nanotubes—How Pericytes Control Blood Flow
Quick Links
In the healthy central nervous system, blood flows where neurons need it most. Now, researchers led by Adriana Di Polo at the University of Montreal help explain why. In the August 12 Nature, they describe a new mechanism for fine control of neurovascular coupling in the retinae of living mice. Delicate tubes connecting pericytes on adjoining capillaries coordinate regional blood flow, they find.
- Tunneling nanotubes connect pericytes on different capillaries in mouse retinae.
- In response to light, one pericyte constricts its capillary, the other widens.
- The coordinated action depends on calcium waves traveling through the nanotube.
These thread-like structures looked like tunneling nanotubes. They extended from one cell, formed a gap junction at the distal end with another, and were packed with cytoplasm and organelles. Calcium waves propagated in either direction through the tubes, suggesting the linked pericytes were communicating. In response to neuronal activation, nearby pericytes relaxed to let blood vessels dilate, while their distal, connected partners contracted, constricting blood flow where it was not needed. This coordinated but opposing action may help channel blood in the retina, where supply is limited, to the most active neurons, the authors believe. “This is the first time a physiological role for any tunneling nanotube has been demonstrated in vivo,” Di Polo told Alzforum.
Chiara Zurzolo at the Pasteur Institute in Paris called the findings significant, and the morphological and functional evidence for the structures impressive. “By using state-of-the-art imaging approaches, Di Polo and collaborators provide the first evidence for the existence of closed tunneling-nanotube-like protrusions in the retina of living mice,” Zurzolo wrote to Alzforum. She also wrote a News & Views for the paper.
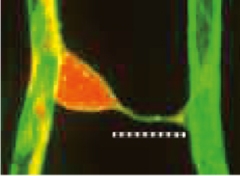
Mysterious Connection. A tunneling nanotube (above dotted line) surrounded by basement membrane (green) connects the cell body of a proximal pericyte (red) to the process of a distal pericyte on another capillary. [Courtesy of Alarcon-Martinez et al., Nature.]
Tunneling nanotubes were first described in cell culture by Hans-Hermann Gerdes and colleagues at the University of Heidelberg, Germany. These slender ducts, around 200 nm in diameter, transferred organelles between cells (Rustom et al., 2004; Gerdes et al., 2007). Other studies reported that nanotubes coordinate electrical signaling among cultured cells (Wang et al., 2010). However, researchers have debated whether the structures exist in vivo, and what purpose they serve.
Separately, the literature as far back as 1838 contains reports of fine “vascular strands” linking capillaries in fixed retinal tissue, Di Polo said. More recent studies of fixed mouse retinae note the existence of thin “bridging” filaments connecting pericytes on neighboring capillaries (e.g., Mendes-Jorge et al., 2012). But no one had characterized these filaments in living tissue.
Joint first authors Luis Alarcon-Martinez and Deborah Villafranca-Baughman were studying mice whose pericytes are fluorescently labeled when they noticed the fine strands linking these cells in the retina. The tubes were about 500 nm in diameter, surrounded by basement membrane, and linked the cell body of one pericyte, the proximal one, with the process of a distal pericyte on another capillary (see image above). A pericyte extends processes far along its capillary, and that is where the distal end of the nanotube attached, not anywhere near the cell body. About two-thirds of the nanotubes were shorter than 30 microns. About a quarter of retinal pericytes had these structures.

Paired Pericytes. An interpericyte tunneling nanotube (IP-TNT, dotted line) connects the cell body (purple) of a proximal pericyte (red) on one capillary to the process of a distal pericyte on another capillary. Other nuclei are blue. [Courtesy of Alarcon-Martinez et al., Nature.]
Electron microscopy of fixed retinas revealed that these tubes were stuffed with material, including mitochondria, vesicles, and endoplasmic reticulum. Structurally, they were supported by an F-actin cytoskeleton and contained a contractile protein called α-smooth muscle actin. The connection with the distal pericyte appeared to be a gap junction, a place where two cell membranes butt up against each other, allowing ions and small molecules to pass between them through narrow channels. Supporting this, the connection contained the gap-junction protein connexin 43, and only small molecules were able to diffuse through; organelles and large molecules were not. Adding a gap-junction blocker prevented this exchange. Mitochondria clustered at the gap junction, hinting at a demand for energy there.
Based on these properties, the authors named the structures interpericyte tunneling nanotubes (IP-TNTs). Di Polo noted that they are distinct from the “bridging pericytes” sometimes seen in the retina when a small blood vessel regresses during vascular remodeling. Those leftover connections are thicker, typically several microns in diameter, and are either empty sleeves of basement membrane or are filled with debris and cell fragments.
To find out what tunneling nanotubes do in the eye, Alarcon-Martinez and colleagues used a two-photon laser scanning microscopy technique they had previously developed to study retinae in living mice. The researchers anesthetized the mice, rotated an eyeball and pulled back a flap of sclera to expose the retina, then placed a small coverslip over it. In mice expressing a fluorescent calcium indicator in their pericytes, the authors saw calcium waves traveling in both directions along IP-TNTs. When the researchers stimulated the retina with light, triggering neuronal activity, one of the linked capillaries constricted a few seconds later, while the other dilated. Notably, calcium levels spiked in pericytes on constricting capillaries, and dropped in those on dilating vessels.
“The observation that one vessel constricts and the other dilates was the most unexpected finding in this study,” Di Polo told Alzforum. She noted that reviewers were skeptical at first, because neuronal activity is believed to increase blood flow, not decrease it. Others may have missed this phenomenon because the changes in blood flow are minute, meaning studies in ex vivo tissue would lack the sensitivity to detect them, Di Polo noted. “When you see it in vivo in thousands of pericyte pairs, it’s very convincing and very strong,” she told Alzforum. In the editorial, Zurzolo said it would be interesting to explore whether IP-TNTs mediate neurovascular coupling over larger brain areas.
Costantino Iadecola at Weill Cornell Medical College in New York told Alzforum that if this same mechanism operates in brain, it could help solve the longstanding mystery of how blood is shunted to only the region that needs it after neuronal activation. “This is fantastic in terms of introducing a new concept to explain how this highly interconnected capillary network could be regulated,” he wrote (full comment below).
Is the calcium wave propagation essential for this regulation? The authors disrupted IP-TNTs in living mice with a tightly focused laser on low power, which did not harm surrounding cells. Afterward, the previously linked pericytes no longer synchronized their constriction and dilation, and blood flow no longer changed after light stimulation. Other health conditions had a similar deleterious effect on nanotubes. For example, when the researchers caused transient ischemia in the retina, calcium levels in pericytes shot up, capillaries constricted, and many nanotubes ruptured. Even after blood flow was restored, these damaged areas were no longer able to alter blood flow in response to light.
Importantly, blocking calcium influx into pericytes just before the ischemia prevented damage to IP-TNTs, and they responded normally to light after reperfusion. “Calcium homeostasis is critical to preserving IP-TNT structure and function,” the authors concluded.
These pericyte tendrils are not confined to the retina. The authors found IP-TNTs in mouse visual cortex and other brain regions as well. In ongoing research, they are looking for IP-TNTs in retinal and brain tissue from monkeys and humans.
Martin Lauritzen at the University of Copenhagen, Denmark, noted that the field of brain pericyte physiology is still developing. “Alarcon-Martinez and colleagues have now identified IP-TNT as a potential mechanism that we will need to take into account in further studies of cerebrovascular function and neurovascular coupling in health and disease,” he wrote to Alzforum (see full comment below).
Could IP-TNTs play a role in neurodegenerative disease? To test this, Di Polo and colleagues are infusing Aβ into living mouse retinae to see what effect it might have on nanotubes and pericyte function. Pathogenic proteins such as misfolded prion and α-synuclein have been shown to travel through tunneling nanotubes in other cell types, hinting the structures may help spread pathology (Gousset et al., 2009; Dec 2016 conference news; Dieriks et al., 2017). Aβ deposits have been found in the retinae of AD patients (Jul 2014 conference news; Sep 2017 news; Apr 2019 conference news).—Madolyn Bowman Rogers
References
News Citations
- Astrocytes and Exosomes Implicated in Protein Propagation
- Alzheimer’s Disease: In the Eye of the Patient?
- Retinal Plaques May Enable Noninvasive Screening for AD
- ARIAS: Zooming in On Eye-based Biomarkers for Alzheimer’s
Paper Citations
- Rustom A, Saffrich R, Markovic I, Walther P, Gerdes HH. Nanotubular highways for intercellular organelle transport. Science. 2004 Feb 13;303(5660):1007-10. PubMed.
- Gerdes HH, Bukoreshtliev NV, Barroso JF. Tunneling nanotubes: a new route for the exchange of components between animal cells. FEBS Lett. 2007 May 22;581(11):2194-201. Epub 2007 Apr 4 PubMed.
- Wang X, Veruki ML, Bukoreshtliev NV, Hartveit E, Gerdes HH. Animal cells connected by nanotubes can be electrically coupled through interposed gap-junction channels. Proc Natl Acad Sci U S A. 2010 Oct 5;107(40):17194-9. Epub 2010 Sep 20 PubMed.
- Mendes-Jorge L, Llombart C, Ramos D, López-Luppo M, Valença A, Nacher V, Navarro M, Carretero A, Méndez-Ferrer S, Rodriguez-Baeza A, Ruberte J. Intercapillary bridging cells: immunocytochemical characteristics of cells that connect blood vessels in the retina. Exp Eye Res. 2012 May;98:79-87. Epub 2012 Mar 29 PubMed.
- Gousset K, Schiff E, Langevin C, Marijanovic Z, Caputo A, Browman DT, Chenouard N, de Chaumont F, Martino A, Enninga J, Olivo-Marin JC, Männel D, Zurzolo C. Prions hijack tunnelling nanotubes for intercellular spread. Nat Cell Biol. 2009 Mar;11(3):328-36. Epub 2009 Feb 8 PubMed.
- Dieriks BV, Park TI, Fourie C, Faull RL, Dragunow M, Curtis MA. α-synuclein transfer through tunneling nanotubes occurs in SH-SY5Y cells and primary brain pericytes from Parkinson's disease patients. Sci Rep. 2017 Feb 23;7:42984. PubMed.
Further Reading
Primary Papers
- Alarcon-Martinez L, Villafranca-Baughman D, Quintero H, Kacerovsky JB, Dotigny F, Murai KK, Prat A, Drapeau P, Di Polo A. Interpericyte tunnelling nanotubes regulate neurovascular coupling. Nature. 2020 Aug 12; PubMed.
- Zurzolo C. Evidence that tunnelling nanotube-like structures connect cells in mice. Nature. 2020 Aug 12; PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University of Copenhagen and Rigshospitalet
University of Copenhagen
Neurovascular coupling by capillary inter-pericyte tunneling nanotubes—a new player in cerebrovascular physiology?
Brain pericyte physiology is an emerging field that underlines how our understanding of brain cerebrovascular function, despite great progress, is limited. The field is still developing and the debate about whether capillary pericytes are contractile or not has been ongoing for more than 100 years (Krogh, 1919). The debate was reignited by a paper from Peppiatt et al. in 2006 that described contractile “bump-on-a-log” pericytes in brain slices. The debate has been muddied by a disagreement on mural cell nomenclature, where some scientists have moved away from the original definition set by Zimmermann and created their own terminology and named all mural cells that express α-smooth muscle actin (α-SMA) vascular smooth muscle cells (VSMC), while non-α-SMA expressing mural cells are termed pericytes (Hill et al., 2015). The point is that all non-spindle-shaped morphological mural cells on intraparenchymal arterioles and capillaries that are not true VSMCs should be named pericytes. While the contractile ability of ensheathing pericytes on first- to fourth-order capillaries are now generally accepted (Grant et al., 2019; Cai et al., 2018; Grubb et al., 2020), the contractility of α-SMA-lacking true pericytes on fifth-plus-order capillaries is still a matter of debate (Hill et al., 2015; Hartmann et al., 2020; Alarcon-Martinez et al., 2019; Grutzendler and Nedergaard, 2019). A previous study by Alarcon-Martinez et al. showed that α-SMA may be lost during retinal fixation if actin depolymerization is not prevented (Alarcon-Martinez et al., 2018) and that this source of error may explain different views of pericyte function.
This new Nature paper by Alarcon-Martinez et al. brings more wood to the fire (Alarcon-Martinez et al., 2020) by presenting a putatively new feature of neurovascular coupling—interpericyte tunneling nanotubes (IP-TNT). TNTs are nanometer-sized tubes that connect the cytoplasm of two distant cells (Korenkova et al., 2020). In their new paper, the authors use the term to describe pericyte processes that do not form cytoplasmic connections, but gap junction connections. IP-TNT’s previously have been described in isolated pericytes where they conveyed α-synuclein transfer from one cell to another by budding off a large vesicle from the end of the TNT as the cells moved apart (Dieriks et al., 2017). The lack of cytoplasmic connections between the pericytes may suggest that it is more accurate to call them “processes of bridging pericytes.” Bridging pericytes are described in the recent preprint by Hartmann et al., who reported that ablation of bridging pericytes dilate both the proximal and distal capillaries, which supports their claim that true pericytes exert a tonus on the capillaries (Hartmann et al., 2020). We have observed bridging pericytes in the whisker barrel cortex, but only in young adults (12-14 weeks), which may suggests that some of the pericyte processes represent a remnant of vascular pruning during development (Korn and Augustin, 2015).
Alarcon-Martinez et al. argue against the pruning idea by referring to (i) the lack of endothelial markers, (ii) α-SMA staining in the IP-TNT, (iii) presence of organelles and mitochondria, (iv) process stability, and (v) existence of intercellular Ca2+ waves. Furthermore, the paper reports IP-TNTs in 6-month-old mice. It would be interesting to develop a method that quantifies IP-TNTs and to examine whether the IP-TNT density changes with age, which is important because IP-TNTs are the morphological substrate for interpericyte calcium signals. A recent preprint by Glück et al. shows how VSMC and ensheathing pericytes synchronize Ca2+ fluctuations, but Ca2+ fluctuations of true pericytes are irregular (Glück et al., 2020), which makes it difficult to interpret the relevance of intercellular Ca2+ waves between pericytes in terms of neurovascular coupling. One could argue that for an intercellular Ca2+ wave between pericytes to be relevant for blood flow control, it would need to travel from the ensheathing pericytes at the first- to fourth-order capillaries, which are crucial for blood flow control in the microvessels, to the precapillary sphincter and the penetrating arteriole Grubb et al., 2020). Future studies will need to address these and other issues. However, Alarcon-Martinez and colleagues have now identified IP-TNT as a potential factor that we will need to take into account in further studies of cerebrovascular function and neurovascular coupling in health and disease.
References:
Krogh A. The supply of oxygen to the tissues and the regulation of the capillary circulation. J Physiol. 1919 May 20;52(6):457-74. PubMed.
Peppiatt CM, Howarth C, Mobbs P, Attwell D. Bidirectional control of CNS capillary diameter by pericytes. Nature. 2006 Oct 12;443(7112):700-4. Epub 2006 Oct 1 PubMed.
Zimmermann KW. Der feinere Bau der Blutcapillaren. Anat Entwicklungsgesch, 1923
Hill RA, Tong L, Yuan P, Murikinati S, Gupta S, Grutzendler J. Regional Blood Flow in the Normal and Ischemic Brain Is Controlled by Arteriolar Smooth Muscle Cell Contractility and Not by Capillary Pericytes. Neuron. 2015 Jun 23; PubMed.
Grant RI, Hartmann DA, Underly RG, Berthiaume AA, Bhat NR, Shih AY. Organizational hierarchy and structural diversity of microvascular pericytes in adult mouse cortex. J Cereb Blood Flow Metab. 2019 Mar;39(3):411-425. Epub 2017 Sep 21 PubMed.
Cai C, Fordsmann JC, Jensen SH, Gesslein B, Lønstrup M, Hald BO, Zambach SA, Brodin B, Lauritzen MJ. Stimulation-induced increases in cerebral blood flow and local capillary vasoconstriction depend on conducted vascular responses. Proc Natl Acad Sci U S A. 2018 Jun 19;115(25):E5796-E5804. Epub 2018 Jun 4 PubMed.
Grubb S, Cai C, Hald BO, Khennouf L, Murmu RP, Jensen AG, Fordsmann J, Zambach S, Lauritzen M. Precapillary sphincters maintain perfusion in the cerebral cortex. Nat Commun. 2020 Jan 20;11(1):395. PubMed.
Alarcon-Martinez L, Yilmaz-Ozcan S, Yemisci M, Schallek J, Kılıç K, Villafranca-Baughman D, Can A, Di Polo A, Dalkara T. Retinal ischemia induces α-SMA-mediated capillary pericyte contraction coincident with perivascular glycogen depletion. Acta Neuropathol Commun. 2019 Aug 20;7(1):134. PubMed.
Grutzendler J, Nedergaard M. Cellular Control of Brain Capillary Blood Flow: In Vivo Imaging Veritas. Trends Neurosci. 2019 Aug;42(8):528-536. Epub 2019 Jun 26 PubMed.
Alarcon-Martinez L, Yilmaz-Ozcan S, Yemisci M, Schallek J, Kılıç K, Can A, Di Polo A, Dalkara T. Capillary pericytes express α-smooth muscle actin, which requires prevention of filamentous-actin depolymerization for detection. Elife. 2018 Mar 21;7 PubMed.
Alarcon-Martinez L, Villafranca-Baughman D, Quintero H, Kacerovsky JB, Dotigny F, Murai KK, Prat A, Drapeau P, Di Polo A. Interpericyte tunnelling nanotubes regulate neurovascular coupling. Nature. 2020 Aug 12; PubMed.
Korenkova O, Pepe A, Zurzolo C. Fine intercellular connections in development: TNTs, cytonemes, or intercellular bridges?. Cell Stress. 2020 Jan 7;4(2):30-43. PubMed.
Dieriks BV, Park TI, Fourie C, Faull RL, Dragunow M, Curtis MA. α-synuclein transfer through tunneling nanotubes occurs in SH-SY5Y cells and primary brain pericytes from Parkinson's disease patients. Sci Rep. 2017 Feb 23;7:42984. PubMed.
Korn C, Augustin HG. Mechanisms of Vessel Pruning and Regression. Dev Cell. 2015 Jul 6;34(1):5-17. PubMed.
Weill College Medicine
I was really fascinated by this paper. The field has always been struck by the restricted nature of the increases in blood flow produced by neural activity. For example, if you mechanically stimulate a mouse whisker, only the arteriole that goes to that region of the barrel cortex dilates. How is that possible, when the capillary network is interconnected like the streets in downtown Boston? We didn’t have the right instruments to sort that out.
Now, Alarcon-Martinez and colleagues report that when two pericytes in the retina are interconnected by a tunneling nanotube, one dilates its vessel and the other constricts in response to neural activity. Assuming this mechanism also operates in brain, that would provide a hemodynamic way to focalize the increase in flow into certain areas. This is fantastic in terms of introducing a new concept to explain how this highly interconnected capillary network could be regulated. Capillaries are the ultimate oxygenation machine in the brain, but their regulation has been underexplored. This paper puts the focus back on capillary hemodynamics.
Make a Comment
To make a comment you must login or register.