New Tracer May Prove Better for Amyloid PET
Quick Links
There are three FDA-approved PET ligands on the market for detecting Aβ by brain imaging, but according to Vijay Sharma, they have major shortcomings. They bind nonspecifically to white matter, and they are blind to diffuse plaques, which may occur earlier in the disease than the dense-core variety. Sharma and his colleagues at Washington University School of Medicine, St. Louis, have created a new Aβ ligand to overcome these limitations, and they introduce it in the November 2 Nature Scientific Reports.
Like the approved troika—florbetapir, florbetaben, and flutemetamol—the new tracer, dubbed fluselenamyl, also relies on a fluorine 18 radioisotope, but its low binding to white matter and high affinity for diffuse plaques renders it both more specific and more sensitive, according to the authors. “Given the interest in very early detection, it is exciting to have the ability to measure diffuse plaques and understand whether they inform us about risk of future dementia,” said Beth Mormino, Massachusetts General Hospital. She was not involved in the work.
New Tracer.
After injection into tail vein, fluselenamyl (green) binds Aβ in brain plaques and blood vessels (red). [Courtesy of Sharma et al., Scientific Reports.]
The researchers focused on a novel class of F18 Aβ tracers that substitute selenium for sulphur inside a benzothiazole (C7H5NS); this widens the diameter of the benzothiazole ring. The paper contains the scientists’ nine-step recipe for synthesizing fluselenamyl.
In postmortem frontal cortex slices from people with confirmed AD, the authors found that fluselenamyl binds selectively to plaques in gray matter while leaving white matter largely untouched. What’s more, fluselenamyl’s octanol/water partition coefficient—a proxy for white matter affinity—is on a par with that of C11 Pittsburgh Compound B. PiB is the gold standard Aβ PET tracer used in research settings, according to David Knopman, Mayo Clinic, Rochester, Minnesota. Knopman explained that C11 PiB has a sufficiently high signal-to-noise ratio to quantify Aβ with PET, but because the C11 radioisotope has a half-life of only 20 minutes, commercial applications have focused on the more stable F18.
To test whether fluselenamyl labels Aβ plaques in a living animal, the researchers injected it into 15-month-old APP/PS1 transgenic mice, which have accumulated robust amyloid plaques by that age. Fifteen to 30 thirty minutes later, the mice brains lit up in the microPET/CT scanner. Although the transgenic mice retained only 20 percent more ligand than did the wild-type mice, this is a big advance over other tracers that show no difference between APP/PS1 mice and controls, Sharma said. Moreover, PET resolution in mice is low and the need for anesthesia slows pharmacokinetics, so Sharma expects fluselenamyl to produce a stronger signal in human PET.

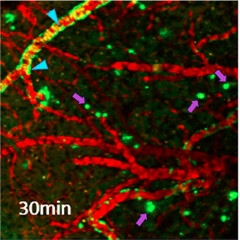
Micro PET.
After intravenous injection, more F18-fluselenamyl binds in the cortex (purple arrows) of APP/PS1 (bottom) than wild-type mice (top). Cerebellum (red arrowhead) accumulates almost no tracer. [Courtesy of Sharma et al., Scientific Reports.]
In the APP/PS1 brains, regional differences in fluselenamyl binding were readily apparent (see image at left), with little binding in the cerebellum and most of the ligand accumulating in the cortex. Such differences have not been seen in mouse brain using PiB or flutemetamol, note the authors. In hippocampal brain slices from the transgenics, fluselenamyl bound to both diffuse and compact plaques, but the researchers did not directly compare to PiB or flutemetamol binding.
Although the authors claim that fluselenamyl detects diffuse plaques better than other ligands do, Chet Mathis, University of Pittsburgh Medical Center, points out that the authors did not directly compare the performance of their compound to that of any other tracer, including the experimental ligand F18 NAV4694. Many in the PET field consider NAV4694 to be the best tracer yet, but its clinical development has been frustratingly slow (see May 2010 news). Without evidence that fluselenamyl outperforms available ligands, the advantage of this new compound for basic research is unclear, Mathis said. He also questioned whether there is a need for another F18 Aβ imaging agent, given that there are three approved and NAV4694 waiting in the wings.
Furthermore, according to Mathis, fluselenamyl has a major drawback. In mice, bone tissue takes up quite a lot of it. That could be a problem in the clinic, because artifacts from the skull could overshadow signal from the brain. None of the other F18 Aβ ligands has this problem.
Experts in the field agree that it is premature to speculate on whether fluselenamyl will be useful until clinical trials are done in humans. Sharma expects that to begin in two years.
“Human studies are the litmus test. They can be humbling even when the preclinical data is as promising as this,” William Klunk of the University of Pittsburgh Medical Center told Alzforum. “This tracer will have to blow the other tracers away in human studies to make the field take notice, and that will be a tough task.”
If clinical trials are successful, Mormino sees potential for this compound, at least in research settings. For one, it could shed light on how diffuse plaques play into aging and AD. Diffuse plaques are very common and whether they matter for AD above and beyond the dense plaques is unknown, she said. Without a specific marker for diffuse plaques, researchers haven’t been able to examine their role in vivo.
For another, Mormino would like to be able to use a more sensitive Aβ PET ligand to differentiate early AD from those without amyloid pathology. Fluselenamyl might reveal whether people with mild symptoms but negative amyloid PET scans are truly negative, or if the current tracers simply miss their diffuse amyloid deposits. “We might be able to detect more people who are at risk for AD very early on using a ligand like [fluselenamyl],” Mormino said.—Erin Hare
Erin Hare is a freelance writer based in Pittsburgh.
References
Research Models Citations
News Citations
Further Reading
No Available Further Reading
Primary Papers
- Sundaram GS, Dhavale DD, Prior JL, Yan P, Cirrito J, Rath NP, Laforest R, Cairns NJ, Lee JM, Kotzbauer PT, Sharma V. Fluselenamyl: A Novel Benzoselenazole Derivative for PET Detection of Amyloid Plaques (Aβ) in Alzheimer's Disease. Sci Rep. 2016 Nov 2;6:35636. PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.