Therapies Take Aim at Tau
Quick Links
Researchers continue to target tau in hopes of treating patients with Alzheimer’s or other tauopathies. At the Alzheimer’s Association International Conference 2014 held July 12-17 in Copenhagen, Denmark, scientists presented their latest attempts to take aim at misfolded forms of this microtubule-binding protein. Potential therapies included two active vaccines, an antibody, an inhibitor of the enzyme that removes sugar molecules from tau, and an anti-aggregating compound. Most scientists are planning, or have already started, tests in humans.
An Immunological Approach
Some groups are working to harness immunological proteins to tame tau. Maria Pihlgren, AC Immune, Lausanne, Switzerland, presented a poster about the company’s active, liposome-based vaccine. It comprises a synthetic tau peptide attached to a linker that is bound to a lipid bilayer (see Hickman et al., 2011, and Jun 2012 conference story). This group tested various synthetic fragments that contain known pathological phosphorylation sites on tau to see which would generate a vaccine that best slows the progression of tau pathology in mice.
Pihlgren and colleagues tested the various vaccines in wild-type or tau transgenic mice that carried either the V337M/R406W or P301L mutations. They gave each three injections of the liposomal p-tau vaccines over three months. The tau antigens included Y18, S404/S409, S202/T205, T212/S214, and S396/S404 phosphorylation motifs. At the end of the treatment period, the scientists used an ELISA to measure the antibody response. Vaccines that targeted S202/T205, T212/S214, and S396/S404 elicited the strongest responses, but only the one aimed at S396/S404—called ACI-35—reduced aggregated tau in mice that carried the P301L mutation.
In treated mice, the ACI-35 vaccine helped animals retain body weight and delayed the onset of abnormal motor control. They also lived longer, with three times as many untreated mice dying by day 90. Having completed safety and efficacy data in mice (see Theunis et al., 2013), Pihlgren and colleagues began a Phase 1 trial in AD patients in December 2013. This is the first p-tau specific vaccine to be tested in humans. Andrea Pfeifer, CEO of AC Immune, told Alzforum that the company hopes to release data on the trial by the end of this year.
Another active vaccine is under development by scientists led by Michal Novak at Axon Neuroscience, Bratislava, Slovak Republic. Novak maintains that tau is truncated before it is hyperphosphorylated, and claims this cleavage step is therefore the root cause of neurofibrillary tangles in AD (see Feb 2013 news story). His company’s vaccine, AADvac1, targets this truncated, misfolded, pathological form of tau. It consists of a synthetic peptide derived from a tau protein sequence, which is coupled to keyhole limpet hemocyanin, a large antigen commonly used as an immune adjuvant. Novak declined to reveal the exact epitope. He presented safety and efficacy data from animals and preliminary results from a Phase 1 trial.
AADvac1 stopped tau phosphorylation and reduced neurofibrillary tangles in transgenic rodent models of Alzheimer’s disease, Novak reported. He uses a transgenic rat model that expresses truncated human tau. In preclinical safety testing in wild-type rats, rabbits, and dogs, no adverse effects occurred with acute doses or repeat injections for up to 26 weeks, Novak said. A Phase 1 study began last year in Austria. Thirty patients were slated to receive either placebo or vaccine for three months. All will then have the option to receive vaccine in a three-month, open-label extension. The scientists will monitor safety with magnetic resonance brain imaging, clinical and neuropsychiatric observation, cognitive testing, electrocardiography, blood biochemistry, hematology, coagulation measurement, and urine analysis. They will continue to observe patients in a separate 18-month follow-up trial. The company is planning to initiate a 24-month Phase 2 trial in patients with mild AD in early 2015.
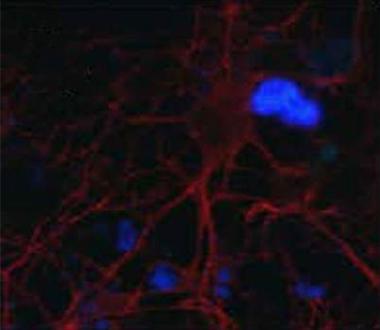
Paired helical filaments (blue) accumulate outside of neurons (red) when antibody is applied at the same time (see text below). [Image courtesy of Erin Congdon and Einar Sigurdsson.]
“Active immunization is a valid approach but riskier than injecting antibodies,” said Einar Sigurdsson at New York University Langone Medical Center, who thought that as long as there are no safety issues, such therapy would have more widespread use than passive immunization as it is cheaper to make and requires fewer injections.
Taking the passive antibody approach, Erin Congdon of Sigurdsson’s lab presented new findings about the tau antibody 4E6G7 (see Jun 2011 conference story). Neurons take up this antibody by clathrin-dependent Fcγ receptor endocytosis, whereupon it helps clear tau (see Congdon et al., 2013). Congdon reported that the antibody prevents uptake of paired helical filaments (PHFs) of tau into neurons. She grew primary neurons from mice in culture and incubated them with PHFs. In some cells she added antibody simultaneously, and in others applied it 24 hours later. In the latter group, antibody entered the cell and bound to tau aggregates. In the former, PHFs remained outside the cell, where they associated with the antibody. The findings suggest that the antibody formed complexes with the PHFs that prevented their uptake, said Sigurdsson.
Tau is thought to spread from neuron to neuron (see Aug 2013 conference story). To see the effect on spread between cells, Congdon cultured neurons on a special plate divided into two parts by a barrier that allowed only axons to pass through. She bathed the neurons on one side with PHFs with or without antibody. The untreated cells took up the tau fragments, which spread via the axons to the neurons on the other side. However, if 4E6G7 was applied at the same time or even 24 hours later, fewer PHFs spread to the neurons at the other end of the plate. “We think that the antibodies are acting intracellularly and extracellularly,” Sigurdsson told Alzforum, saying that multiple mechanisms are at play to prevent the spread of tau from cell to cell.
Disrupting Aggregation
Other scientists are working on therapies that prevent or break up the clumping of tau. Dirk Beher, Asceneuron, Lausanne, Switzerland, talked about ASN-561, his company’s inhibitor of O-GlcNAcase. This glycoside hydrolase removes sugars from tau and similarly modified proteins. Other groups also have adopted this strategy (see Mar 2012 news story). ASN-561 leaves more sugars on tau and makes it less likely to aggregate, reducing neurofibrillary tangles, explained Beher.
Because tau O-GlcNAcylation, or the process of adding sugars, decreases both with age and in Alzheimer’s disease, researchers believe that preventing the removal of sugars may restore tau's coat to a more youthful appearance (see Liu et al., 2009). Previously, they found that the inhibitor Thiamet G reduced O-GlcNAcase activity and prevented tangles from forming in tau transgenic mice (see Yuzwa et al., 2012). However, high doses were needed since the compound enters the brain poorly, said Beher. Asceneuron screened for an inhibitor that better entered the CNS, he said. After optimizing one of the hits, the group developed ASN-561, which potently inhibits O-GlcNAcase and crosses the blood-brain barrier.
To examine how it affected tau buildup, the researchers treated JNPL3 mice with ASN-561 for five days. They monitored O-GlcNAcylated tau with an antibody that specifically recognizes the O-GlcNAcylated form of the protein (see Cameron et al., 2013). In the forebrain, brain stem, and spinal cord of treated mice, the levels of O-GlcNAcylated tau rose dose-dependently up to 12-fold higher than they did in vehicle-treated mice, suggesting the compound hits the right target. So far, scientists have observed no safety risks with the compound, Beher said. Studies of tau aggregation and behavior are next.
If preclinical development is successful, the group plans to treat people with progressive supranuclear palsy, a rare, fast-progressing tauopathy. The hope is that PSP might lead to faster approval than if they started with Alzheimer’s disease. Beher said they are on track to begin the first human studies in late 2015.
Audience members asked about improvements in mice that express wild-type tau. Beher responded that he has not evaluated the compound in wild-type animals, given that they produce no neurofibrillary tangles. Others wondered if O-GlcNAcase knockouts are viable. Knockouts die before birth, Beher said. Pharmacological inhibition seems less drastic since Thiamet G had no major effects when given to mice for up to six months, he said. “We are not trying to inhibit the enzyme fully, just change the balance of O-GlcNAcylation to a healthier state,” said Beher.
While this is an interesting approach and the compound looks promising, these are still early days, said Eric Karran, Alzheimer’s Research UK, Cambridge. He commented that this is an example of an enzyme that has few known homologues and yet inhibiting it seems to have few toxic effects. “So far it looks like the body can tolerate ASN-561 pretty well,” he told Alzforum. Long-term toxicology tests will further explore that possibility. The group will also need to demonstrate that the compound attenuates tau pathology, Karran said. So far, scientists have struggled to tackle tau toxicity. “When it comes to affecting tau, there are not that many mechanisms that you can go after, so the more diversity of tau targets we have, the better.”
Jens Wagner, now at the German Center for Neurodegenerative Diseases (DZNE), Bonn, Germany, presented data on a general oligomer buster called anle138b. Wagner formerly worked with Armin Giese, Ludwig-Maximilians-University Munich. Giese’s group previously reported that anle138b guards against prion and α-synuclein oligomer accumulation in mouse models of prion and Parkinson’s diseases, respectively, and prevents neurodegeneration and disease progression (see April 2011 conference story and Wagner et al., 2013).
To see if anle138b works in animal models of tauopathy, Wagner and colleagues gave the compound to tau transgenic mice. These animals overexpress human P301S tau, deposit the hyperphosphorylated protein in the cerebral cortex, hippocampus, brain stem, and spinal cord, and lose synapses and neurons, especially in the spinal cord, as they age. Treatment starting at weaning reduced the number of tau aggregates that formed before the animals died, according to Western blots. It also partially lowered synapse and neuron loss in the CA3 region and in the stratum lucidum of the hippocampus, according to stains for NeuN and synaptophysin. Treated mice performed better than controls in a novel object recognition learning and memory task. They also lived slightly longer—about two months—than untreated animals, though both survived significantly fewer months than wild types.
Giese and colleagues founded the company MODAG GmbH, which has licensed anle138b for further development. “Our hope is that next year we can start clinical studies,” said Giese. After preclinical safety testing, the group will start Phase 1 trials in healthy volunteers, and, if all goes well, Giese said they will move to Phase 2 in a population of people with a rare disease such as multiple system atrophy, a rapidly progressing synucleinopathy. These patients deteriorate quickly, which would allow scientists to measure outcomes in a relatively short timespan.
“Wagner provided extensive histological, neuropathological, and behavioral data for P301S mice treated with this drug,” said Eugenia Trushina, Mayo Clinic, Rochester, Minnesota, who chaired the session. “In combination with published work, I think they have solid data to support their therapeutic strategy.” Co-chair Thomas Wisniewski, New York University Langone Medical Center, agreed. “Aggregation is a nice target and he presented good data,” he told Alzforum. However, Wisniewski cautioned that the scientists started treatment in these mice before any misfolded tau had accumulated, so this represented a preventive strategy. He wondered if it would work as well after tau tangles had accumulated. By the time people with tauopathies are symptomatic, they have substantial tau buildup already, he pointed out. He also noted that the potential toxicity of anle138b compounds is unknown.—Gwyneth Dickey Zakaib
References
News Citations
- Stockholm: New Strategies for Immunotherapy
- Truncated Tau Triggers Tangles, Transmits Pathology
- Uppsala: Is Tau Immunotherapy Taking Off?
- Tales of Traveling Tau: Is Transfer Between Neurons Normal?
- Can a Little Sugar Keep Tau From Souring Neurons?
- Barcelona: Parkinson’s Treatments on the Horizon
Research Models Citations
Paper Citations
- Hickman DT, López-Deber MP, Ndao DM, Silva AB, Nand D, Pihlgren M, Giriens V, Madani R, St-Pierre A, Karastaneva H, Nagel-Steger L, Willbold D, Riesner D, Nicolau C, Baldus M, Pfeifer A, Muhs A. Sequence-independent control of peptide conformation in liposomal vaccines for targeting protein misfolding diseases. J Biol Chem. 2011 Apr 22;286(16):13966-76. PubMed.
- Theunis C, Crespo-Biel N, Gafner V, Pihlgren M, López-Deber MP, Reis P, Hickman DT, Adolfsson O, Chuard N, Ndao DM, Borghgraef P, Devijver H, Van Leuven F, Pfeifer A, Muhs A. Efficacy and Safety of A Liposome-Based Vaccine against Protein Tau, Assessed in Tau.P301L Mice That Model Tauopathy. PLoS One. 2013;8(8):e72301. PubMed.
- Filipcik P, Zilka N, Bugos O, Kucerak J, Koson P, Novak P, Novak M. First transgenic rat model developing progressive cortical neurofibrillary tangles. Neurobiol Aging. 2012 Jul;33(7):1448-56. Epub 2010 Dec 31 PubMed.
- Congdon EE, Gu J, Sait HB, Sigurdsson EM. Antibody uptake into neurons occurs primarily via clathrin-dependent Fcγ receptor endocytosis and is a prerequisite for acute tau protein clearance. J Biol Chem. 2013 Dec 6;288(49):35452-65. Epub 2013 Oct 25 PubMed.
- Liu F, Shi J, Tanimukai H, Gu J, Grundke-Iqbal I, Iqbal K, Gong CX. Reduced O-GlcNAcylation links lower brain glucose metabolism and tau pathology in Alzheimer's disease. Brain. 2009 Jul;132(Pt 7):1820-32. PubMed.
- Yuzwa SA, Shan X, Macauley MS, Clark T, Skorobogatko Y, Vosseller K, Vocadlo DJ. Increasing O-GlcNAc slows neurodegeneration and stabilizes tau against aggregation. Nat Chem Biol. 2012 Apr;8(4):393-9. PubMed.
- Cameron A, Giacomozzi B, Joyce J, Gray A, Graham D, Ousson S, Neny M, Beher D, Carlson G, O'Moore J, Shearman M, Hering H. Generation and characterization of a rabbit monoclonal antibody site-specific for tau O-GlcNAcylated at serine 400. FEBS Lett. 2013 Nov 15;587(22):3722-8. Epub 2013 Oct 7 PubMed.
- Wagner J, Ryazanov S, Leonov A, Levin J, Shi S, Schmidt F, Prix C, Pan-Montojo F, Bertsch U, Mitteregger-Kretzschmar G, Geissen M, Eiden M, Leidel F, Hirschberger T, Deeg AA, Krauth JJ, Zinth W, Tavan P, Pilger J, Zweckstetter M, Frank T, Bähr M, Weishaupt JH, Uhr M, Urlaub H, Teichmann U, Samwer M, Bötzel K, Groschup M, Kretzschmar H, Griesinger C, Giese A. Anle138b: a novel oligomer modulator for disease-modifying therapy of neurodegenerative diseases such as prion and Parkinson's disease. Acta Neuropathol. 2013 Jun;125(6):795-813. PubMed.
External Citations
Further Reading
Papers
- Castillo-Carranza DL, Sengupta U, Guerrero-Muñoz MJ, Lasagna-Reeves CA, Gerson JE, Singh G, Estes DM, Barrett AD, Dineley KT, Jackson GR, Kayed R. Passive immunization with Tau oligomer monoclonal antibody reverses tauopathy phenotypes without affecting hyperphosphorylated neurofibrillary tangles. J Neurosci. 2014 Mar 19;34(12):4260-72. PubMed.
- Giacobini E, Gold G. Alzheimer disease therapy--moving from amyloid-β to tau. Nat Rev Neurol. 2013 Dec;9(12):677-86. Epub 2013 Nov 12 PubMed.
- Rosenmann H. Immunotherapy for targeting tau pathology in Alzheimer's disease and tauopathies. Curr Alzheimer Res. 2013 Mar;10(3):217-28. PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University of Pennsylvania
These are exciting early studies of active tau immune therapy, but one concern is the possibility that the use of tau peptides as vaccines exposes individuals to the possibility of induction of CNS tau pathology. Peripheral administration of tau has been shown to trigger CNS tauopathy in experimental models (see Clavaguera et al., 2014), and peripheral administration of Aβ (see Eisele et al., 2010) and α-synuclein (Sacino et al., 2014) can trigger CNS Aβ and α-synuclein pathology, respectively. Thus, further studies are needed to be sure this does not happen in patients.
References:
Clavaguera F, Hench J, Lavenir I, Schweighauser G, Frank S, Goedert M, Tolnay M. Peripheral administration of tau aggregates triggers intracerebral tauopathy in transgenic mice. Acta Neuropathol. 2014 Feb;127(2):299-301. Epub 2013 Dec 21 PubMed.
Eisele YS, Obermüller U, Heilbronner G, Baumann F, Kaeser SA, Wolburg H, Walker LC, Staufenbiel M, Heikenwalder M, Jucker M. Peripherally applied Abeta-containing inoculates induce cerebral beta-amyloidosis. Science. 2010 Nov 12;330(6006):980-2. PubMed.
Sacino AN, Brooks M, Thomas MA, McKinney AB, Lee S, Regenhardt RW, McGarvey NH, Ayers JI, Notterpek L, Borchelt DR, Golde TE, Giasson BI. Intramuscular injection of α-synuclein induces CNS α-synuclein pathology and a rapid-onset motor phenotype in transgenic mice. Proc Natl Acad Sci U S A. 2014 Jul 22;111(29):10732-7. Epub 2014 Jul 7 PubMed.
Make a Comment
To make a comment you must login or register.