Tau Tracers Shine at Boston Conference
Quick Links
Ligands that light up tau on brain scans generated plenty of buzz among researchers attending the Alzheimer's Association International Conference, held July 13-18 in Boston. Presenters at the meeting offered the latest data on three compound classes. They are T807 and T808, being developed by Eli Lilly and Company through their subsidiary Avid Radiopharmaceuticals. Then there is the THK serious of quinoline-derived compounds named after Tohoku University in Sendai, Japan. The third, and newest, player is PBB3, a benzothiazole derivative similar to Pittsburgh compound B (PiB). PBB3 is being developed at National Institute of Radiological Sciences in Chiba, Japan.
The number of people who have undergone positron emission tomography (PET) with these tracers is still small—in the dozens thus far. That said, early human data show two promising, crucial characteristics, said William Klunk of the University of Pittsburgh in Pennsylvania. Several of the compounds generate a good signal-to-noise ratio, and they label brain areas where tau pathology occurs. One important step to validate the ligands, said Klunk, will be to formally prove they highlight the same areas of the brain where tau aggregates are found on postmortem examination. Lilly presented early postmortem results from one study participant who died of unrelated causes soon after having a T808 scan.
In addition to postmortem validation, researchers have to answer plenty of questions about these tau ligands before they are ready for prime time. Will the ligands help identify non-AD tauopathies, such as chronic traumatic encephalopathy (CTE), corticobasal degeneration (CBD), or progressive supranuclear palsy (PSP)? Since aggregating tau takes different forms, one cannot assume that one ligand will bind them all, noted William Jagust, University of California in Berkeley, and other PET experts who spoke with Alzforum. One crucial test, Klunk noted, is that the ligands must bind tau, but not Aβ amyloid. This is difficult because the latter is much more abundant in the AD brain and could cause false positive scans unless the ligand has a much higher specific affinity for tau than for Abeta. Nonspecific binding plagued early tau tracers such as FDDNP, which are said to bind both beta amyloid and tau deposits (Small et al., 2013).
Lilly’s Compounds Offer Initial Post-Mortem Data
Lilly acquired the fluorine-tagged ligands T807 and T808 from Siemens Molecular Imaging Biomarker Research in April 2013 (see Alzforum News story). These compounds came out of compound library screens that used human brain slices laden with tau tangles as binding targets. T807/8 have different heteroaromatic core structures, but bind the same pocket of tau with similar affinity (Zhang et al., 2012) .
Thus far, Siemens and Avid researchers have run T807 PET scans of 10 people—three with a low probability of AD and seven with a high probability, based on clinical examination. In his AAIC presentation, Mark Mintun from Avid expanded on previously published data from six of those people (see ARF related news story). The strength of T807 labeling correlated with the severity of cognitive defects experienced by the volunteers, as measured by the Mini-Mental State Exam (MMSE), said Mintun.
One outlier broke the trend. The researchers originally diagnosed this participant with AD, but the scan suggested very little tau pathology. Later, the researchers scanned this same person for Aβ pathology, and that, too, was negative. Mintun concluded the person likely never had AD (see image below).
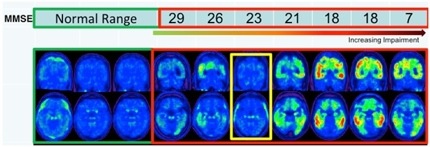
PET imaging with T807: Across normal people and those with likely AD (red box), signal strength increased with worsening scores on the MMSE. One outlier (yellow box) turned out to be amyloid negative. Image courtesy of Mark Mintun, Avid Radiopharmaceuticals, Philadelphia, Pennsylvania.
Mintun concluded that T807 entered the brain and labeled traditional tau-filled regions, including the temporal, parietal, and occipital lobes. The next steps, he said, will be to characterize how those labeled regions correlate with the types of cognitive decline experienced by participants. Hartmuth Kolb, formerly at Siemens but now also at Avid Radiopharmaceuticals, tested T808 in 12 people, eight with cognitive impairment (MCI or AD) and four healthy controls. As with T807, Kolb observed that the worse off a participant was cognitively, the more tau the tracer stained in their brains, with labeling in the frontal, temporal and parietal lobes as well as the hippocampus.
Furthermore, Kolb presented preliminary data on postmortem validation for T808. One 87-year-old man in the study, who had a diagnosis of likely AD, died of a pulmonary embolism two weeks following his scan; he had agreed to donate his brain. Kolb and colleagues stained his brain with the AT100 antibody for phosphorylated tau. They saw that the post-mortem immunostaining of the frontal, temporal and parietal lobes and the hippocampus agreed with the regions lit up by the PET ligand, Kolb said.
Avid has started investigations to determine if these ligands label tauopathies other than AD. The researchers are checking post-mortem brain samples from several tauopathies with the fluorescent marker T557, which has a similar structure to T808 and likely binds the same kinds of aggregated tau. This label lit up cases of Pick’s disease, CTE and frontotemporal dementia. “We should be able to see tau in these brains” with T808, Kolb suggested.
Beyond Kolb and Mintun, PET experts at different academic institutions are beginning to study these tracers. In years past, Avid Radiopharmaceuticals had readily shared AV45/florbetapir with the research community, in effect tapping the field’s collective expertise to study the compound and gather data that helped move it through development quickly.
THK Team Reports Improved Specificity
For their part, research collaborators from Japan and Australia came to Boston with news of the fluorine-tagged THK tracers, including first-generation THK-523 and second-generation THK-5105 and THK-5117. THK-523 also came out of a screen for tau ligands, which identified several quinoline- and benzimidazole-based compounds (Okamura et al., 2005 ). In mice, THK-523 has good affinity for tau fibrils, but not amyloid (Fodero-Tavoletti et al., 2010). Nobuyuki Okamura, of Tohoku University, reported on 20 human subjects. Ten AD patients and 10 healthy controls underwent scans with THK-523. It labeled areas known to accumulate tau paired helical fragments (PHFs), including the temporal, parietal, and orbitofrontal lobes, and the hippocampus of people with AD.
However, THK-523 binds too strongly in white matter, making it hard to differentiate AD and control profiles, said collaborator Victor Villemagne of Austin Health in Melbourne, Australia, in a discussion with Alzforum. Instead, the researchers are now pursuing the new compounds THK-5105 and THK-5117. These share similar structures with 523 but are optimized for higher affinity binding to tau aggregates (Okamura et al., 2013). THK-5105 shows no retention in white matter, making it better suited to distinguish cases and controls, Villemagne said.
Okamura reported on eight people with AD and eight healthy controls who underwent THK-5105 imaging. The labeled areas correlated with known distributions of tau in the AD brain—the hippocampus and neocortex, in particular the inferior temporal cortex—but not with areas where the PiB amyloid tracer bound. As with other ligands, the 5105 binding increased with the severity of the dementia based on MMSE scores. Okamura told Alzforum that preliminary data on THK-5117 indicates it has better pharmacokinetics than THK-5105; however, the researchers will determine which is best after these first studies are complete.
Since the THK compounds bind PHFs, Villemagne said he suspects they will interact with tau not only in AD, but also in Down syndrome and CTE. THK-5105, but not THK-523, showed some interaction with corticobasal lesions, he said. The compounds are insensitive to Pick bodies and the tangles in PSP, Okamura said. Other clinician-scientists expressed great interest in using PET tracers to better understand the different conformations by which tau aggregates and deposits in the different tau diseases. At this point, no researcher interviewed by Alzforum anticipates that there will be a single pan-tau PET tracer for all tauopathies.
Okamura told Alzforum he hopes to perform longitudinal studies of tau tracers in a large group of people with AD, MCI, and preclinical AD. “By the assessment of spatial and temporal pattern of tau deposition, we will understand the role of PHF-tau in AD progression, cognitive decline, and neurodegeneration,” he suggested.
Progress with PBB3
Finally, researchers led by Hitoshi Shimada of Japan’s National Institute of Radiological Sciences have tested PBB3 in 10 healthy controls, five people with MCI, and 13 with AD. PBB3 labeled large areas of the brain, including the medial temporal cortex, hippocampal limbic system, and neocortex. The tracer’s signal was highest and most widespread in the most severe AD cases. It showed no overlap with amyloid beta ligand binding, Shimada claimed. Tau imaging may reflect the severity of cognitive defects better than amyloid scans, Shimada suggested. He declined to say what specific tauopathies PBB3 might label.
PBB3 is structurally similar to PiB, but has a molecular spacer in its middle that makes it bind to tau. Some scientists who spoke with Alzforum cautioned that they would like to see further data clarifying how well PBB3 distinguishes tau from Abeta amyloid. Others noted that in its current form PBB3 might be less widely useful than the other compounds because it relies on carbon-11, which has a much shorter half life than fluorine-18. Users must have an on-site cyclotron for carbon-11 imaging, making the label less practical.
Evolving Imaging
The AD field has long awaited a validated tau tracer. The advent of promising ligands “for the second major molecular pathology of the disease is a very exciting development,” commented Andrew Saykin of Indiana University in Indianapolis. However, he noted that the wait goes on, as the current crop of tracers requires more validation before regular use in the clinic, or even use to follow people’s tau burden in clinical trials.
Validated tau tracers could help researchers answer many questions. “They will clearly define the relative time course of amyloid deposition versus tau,” said Christopher Rowe of Austin Health in Melbourne, Australia. Rowe’s site is testing the THK compounds. Based on tau in cerebrospinal fluid, scientists estimated that tau changes after amyloid, but imaging would confirm whether the same sequence occurs in the brain.
In clinical trials, tau tracers could help scientists identify the right subjects for potential tau interventions, and follow their progress. “This is becoming a much more urgent need because tau is being recognized more as a potential target for new therapeutics. We need a tool to evaluate these drugs,” commented Ralph Nixon of the Nathan Kline Institute in Orangeburg, New York, who moderated a press conference about the topic. Tau PET scans might also help doctors decide when a person should start a future anti-tau therapy.
Eventually, clinicians might be able to use tau imaging to differentiate Alzheimer’s from normal aging or other conditions, researchers suggested. Tau correlates with disease symptoms better than does amyloid, and the latter can also show up in what appears to be normal aging, noted Jagust. Tau starts to show up as cognitive decline becomes imminent, Rowe said, and thus might help doctors predict symptoms better. However, scientists who spoke with Alzforum noted that the challenges in getting amyloid imaging reimbursed by insurers (see ARF related news story) do not bode well for regular clinical use of tau imaging.
The low abundance of tau aggregates in the brain, and its multiple forms, make tracing this protein a much more complicated task than it was for Aβ, said Klunk. Nonetheless, Rowe noted, the field has seen “an explosion of research with tau imaging.” He anticipates lots more news at next year’s AAIC in Copenhagen, Denmark.—Amber Dance.
References
News Citations
- Lilly Buys Tau Tracers From Siemens
- HAI—Spotlight on Tau Tracers at Human Amyloid Imaging Meeting
- Coverage Denial For Amyloid Scans Riles Alzheimer’s Community
Paper Citations
- Small GW, Kepe V, Siddarth P, Ercoli LM, Merrill DA, Donoghue N, Bookheimer SY, Martinez J, Omalu B, Barrio JR. PET Scanning of Brain Tau in Retired National Football League Players: Preliminary Findings. Am J Geriatr Psychiatry. 2013 Jan 1;(21):138e144.
- Zhang W, Arteaga J, Cashion DK, Chen G, Gangadharmath U, Gomez LF, Kasi D, Lam C, Liang Q, Liu C, Mocharla VP, Mu F, Sinha A, Szardenings AK, Wang E, Walsh JC, Xia C, Yu C, Zhao T, Kolb HC. A Highly Selective and Specific PET Tracer for Imaging of Tau Pathologies. J Alzheimers Dis. 2012 Jan 1;31(3):601-12. PubMed.
- Fodero-Tavoletti MT, Okamura N, Furumoto S, Mulligan RS, Connor AR, McLean CA, Cao D, Rigopoulos A, Cartwright GA, O'Keefe G, Gong S, Adlard PA, Barnham KJ, Rowe CC, Masters CL, Kudo Y, Cappai R, Yanai K, Villemagne VL. 18F-THK523: a novel in vivo tau imaging ligand for Alzheimer's disease. Brain. 2011 Apr;134(Pt 4):1089-100. PubMed.
- Okamura N, Furumoto S, Harada R, Tago T, Yoshikawa T, Fodero-Tavoletti M, Mulligan RS, Villemagne VL, Akatsu H, Yamamoto T, Arai H, Iwata R, Yanai K, Kudo Y. Novel 18F-Labeled Arylquinoline Derivatives for Noninvasive Imaging of Tau Pathology in Alzheimer Disease. J Nucl Med. 2013 Aug;54(8):1420-7. PubMed.
External Citations
Further Reading
No Available Further Reading
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.