A Tau Blood Test Tracks With Alzheimer's Neuropathology
Quick Links
Several biomarkers for Alzheimer's disease pick up amyloid plaque and neurofibrillary tangle load as measured by CSF or PET scans. That said, traditionally, the gold standard for gauging pathology has been analyzing brain tissue postmortem. To begin the process of validating plasma markers against tissue neuropathology, Gemma Salvadó of Lund University in Sweden correlated antemortem blood marker concentrations to the extent of Alzheimer’s disease pathology after death. At the Clinical Trials on Alzheimer’s Disease conference, held November 29 through December 2 in San Francisco, she reported that plasma phospho-tau217 best predicted plaques and tangles. It ticked up years before death in people with even the earliest stage of pathology postmortem. This research, posted as a medRxiv preprint, aligns with other CTAD presentations pointing to p-tau217 as an accurate blood marker to detect AD pathology (see Part 9 of this series).
- Plasma p-tau217 tightly correlated with postmortem plaques and tangles.
- It also detected early accumulation of both pathologies.
- P-tau217 measured five years before death predicted future high pathology.
“It really seems that plasma p-tau217 takes it all,” Philip Scheltens of VU University, Amsterdam, said after Salvadó’s presentation.
Salvadó analyzed plasma markers and neuropathological data from 105 participants who had one blood sample collected within five years of death. Forty percent were women, the average age at death was 85. All were from the Arizona Study of Aging and Neurodegenerative Disorders at the Banner Institute in Phoenix. The plasma markers included in this study were p-tau181, p-tau217, p-tau231, Aβ42/40, glial fibrillary acidic protein, and neurofilament light. Cortical and hippocampal amyloid plaques and neurofibrillary tangles were measured using the CERAD score and Braak stage, respectively; 59 had AD pathology, while 46 did not.
While all but plasma NfL correlated with plaque load, p-tau217 was the strongest, with a correlation coefficient of 0.69 out of a perfect 1.0. P-tau181 and Aβ42/40 came next, with coefficients of 0.58 and -0.53, respectively, followed by GFAP at 0.41 and p-tau231 at 0.37.
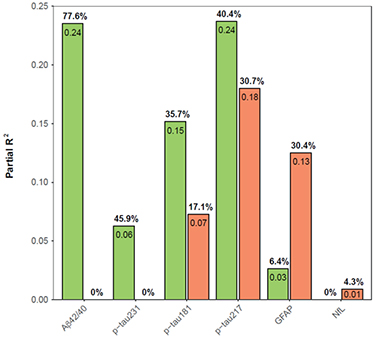
Pathological Contributions. Amyloid plaque load (green) controlled plasma Aβ42/40 and p-tau231 levels, while neurofibrillary tangles (orange) mostly accounted with GFAP changes. Both pathologies contributed to plasma p-tau181 and p-tau217. Neither influenced NfL levels. [Courtesy of Salvadó et al., medRxiv, 2022.]
Similar relationships held for tangles. P-tau217 correlated best among the markers measured in this study, again clocking a coefficient of 0.69. Next came p-tau181 at 0.55, then GFAP at 0.51, and p-tau231 at 0.29. Aβ42/40 was last with a coefficient of -0.27. NfL did not correlate with tangles. Curiously though, NfL did associate with a degradation of cerebral white-matter integrity.
Because the association between a marker and tangles may be influenced by the concomitant presence of plaques, Salvadó was curious how each plasma marker concentration correlated to plaques and tangles independently. To tease this out, she created a multivariable model that calculated the contribution of each pathology individually to each marker concentration. She found that plaque load mostly explained the variance in Aβ42/40 and p-tau231 concentrations, while tangles drove GFAP changes. Both pathologies contributed to p-tau217 and p-tau181 concentration, with plaques having more influence than tangles. Neither pathology controlled NfL.

P-tau217 Predicts Pathology. Plasma p-tau217 collected in the five years prior to death was the strongest individual predictor of plaques and tangles seen postmortem. The parsimonious model includes age, sex, p-tau217, and Aβ42/40. The basic model is age and sex alone. [Courtesy of Salvadó et al., medRxiv, 2022.]
Plasma p-tau217 was the best single marker to predict the presence of both pathologies with an area under the curve of 0.88 (see image below). AUC is a measure of sensitivity and specificity with a perfect score of 1.0. Combining p-tau217 with Aβ42/40 nudged accuracy up to 90 percent.
Did plasma p-tau217 also flag when pathology had first crept up in these people's brains? To answer this, Salvadó assigned each participant an AD neuropathologic change score. The ADNC incorporates CERAD, Braak stage, and Thal amyloid plaque phase to gauge if the person has no, low, intermediate, or high AD pathology.
Indeed, a slight uptick in plasma p-tau217 above values in controls signaled early signs of pathology, flagging people with the lowest ADNC scores in this postmortem sample. Plasma p-Tau217 levels were higher in people with intermediate and high ADNC scores at death (see image below). Differences in the marker between people with no or low pathology and those with intermediate and high pathology were apparent as early as seven years before death in a subset of 48 participants who had multiple blood draws, an average of 3.8 years apart, in their last decade of life.

Earliest of Six. Plasma p-tau217 (top left) detected the earliest accumulation of plaques and tangles seen postmortem in this sample. A drop in plasma Aβ42/40 (bottom left) distinguished people with low from intermediate pathology, while a p-tau181 or p-tau231 (top middle and left) were high only in people with high pathology. GFAP and NfL (bottom middle, left) were uninformative. [Courtesy of Salvadó et al., medRxiv, 2022.]
All told, Salvadó concluded that plasma p-tau217 correlated most strongly with plaques and tangles postmortem. “This marker could be very useful in screening for clinical trials and possibly act as a secondary endpoint due to its longitudinal relationship with pathology,” she said.—Chelsea Weidman Burke
References
News Citations
Further Reading
No Available Further Reading
Primary Papers
- Salvadó G, Ossenkoppele R, Ashton NJ, Beach TG, Serrano GE, Zetterberg H, Mattsson-Carlgren N, Janelidze S, Blennow K, Hansson O. Specific associations between plasma biomarkers and post-mortem amyloid plaque and neurofibrillary tau tangle loads. medRxiv, August 22, 2022. medRxiv
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.