Neuronal Exosomes Embroiled in Controversy
Quick Links
Small snippets of cell membranes that pinch off the neuronal surface are getting swept up in a rather large controversy. Some of these extracellular vesicles wend their way into the cerebrospinal fluid and even plasma, from where they can be plucked and scoured for signs of brain damage. The plucking, however, is a problem, according to researchers led by David Walt, Brigham and Women’s Hospital, Boston. For seven years now, labs around the world have relied on L1CAM, a neuronal cell surface protein, to isolate supposedly neuron-derived extracellular vesicles in biofluids. Alas, in the June 3 Nature Methods online, Walt and colleagues claim L1CAM does not associate with those NDEVs. If they are right, studies that have reported exosome biomarker differences between healthy controls and people with various neurodegenerative diseases, including Alzheimer’s, might be called into question.
- L1CAM sits in neuronal membranes.
- In CSF, plasma, it’s being used to isolate neuronal exosomes.
- New study says this L1CAM is soluble, not associated with vesicles.
Some are not convinced. “The study by Norman et al. makes an important contribution to the ongoing research on deriving neuronal extracellular vesicles from biofluids; however, I cannot accept its bold conclusions regarding L1CAM,” wrote Dimitrios Kapogiannis, National Institute on Aging, Baltimore, to Alzforum. “I think this paper just shows that soluble L1CAM exists in plasma and CSF, something already widely known, but does not show that L1CAM-associated EVs do not exist,” added Kapogiannis, a pioneer in the field of extracellular vesicle (EV) research.
Tsuneya Ikezu, however, thinks the authors are onto something. “The results are convincing and will shed new light on the re-evaluation of the L1CAM-based method for isolating neuron-derived EVs,” Ikezu, now at Mayo Clinic in Jacksonville, Florida, wrote to Alzforum. “Our lab recently studied the protein composition of EVs isolated from iPSC-derived neurons and could not detect L1CAM by mass-spectrometry,” he added.
The issue was debated during a recent workshop organized by the International Society for Extracellular Vesicles and the Michael J. Fox Foundation. One view there held that, for certain research purposes, it may not matter whether L1CAM identifies NDEVs or not. “As a biologist, I would like to know if the L1CAM is coming from neurons, but from a biomarker perspective I think if we have an assay that works and is reproducible, that’s great,” noted Kenneth Witwer, Johns Hopkins University, Baltimore. The idea here is that if L1CAM, however it does so, associates with markers that are informative, it could be useful.
In recent years, L1CAM enrichment has been used to probe exosome changes in AD, Parkinson’s, frontotemporal dementia, multiple systems atrophy, dementia with Lewy bodies, and other neurodegenerative diseases (Shi et al., 2014; Goetzl et al., 2015; Winston et al., 2016; Pulliam et al., 2019; Cha et al., 2019; Jiang et al., 2020).
What’s the new evidence against L1CAM? Co-first authors Maia Norman, Dmitry Ter-Ovanesyan, and colleagues developed highly sensitive SIMOA assays to measure this protein and other vesicle markers in size-exclusion and density-gradient fractions of human CSF and plasma. They found that L1CAM eluted together with soluble albumin, while membrane tetraspanins CD9, CD63, and CD81 eluted with fractions containing larger, lighter structures (see image below). These tetraspanin fractions were enriched for extracellular vesicles as seen in electron micrographs (see image below). In short, L1CAM did not associate with EVs.
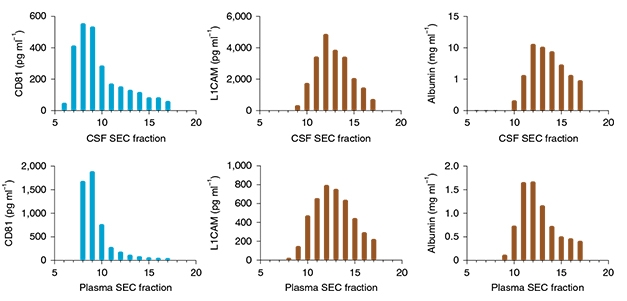
Fractious L1CAM. In size-exclusion chromatography, L1CAM (center) elutes in later fractions along with albumin (right), whereas membrane tetraspanins elute earlier, in keeping with a vesicle origin (left). [Courtesy of Norman et al., Nature Method, 2021.]

Voila, Vesicles. Following size exclusion chromatography of human plasma, a fraction containing tetraspanins (left) is enriched for vesicles, whereas later fractions containing L1CAM and soluble albumin (right) are not. [Courtesy of Norman et al., Nature Methods, 2021.]
L1CAM spans the neuronal cell membrane, so why does it behave as a soluble protein on density gradients and in size-exclusion chromatography? Norman and colleagues think that’s because proteases clip the molecule. CSF L1CAM ran as a 200 kDa protein on western blots and was detectable only with an antibody to the extracellular side of the protein, not one to the intracellular domain. This is consistent with it being the shed extracellular domain, not the full-length 220 kDa protein.
Plasma told a different story, however. There, the soluble protein appeared to be full-length, running as 220 kDa. The authors think this is an alternatively spliced form missing exon 25. Since this exon encodes a small transmembrane domain, this L1CAM would have both extracellular and intracellular domains, but never slide into the membrane. Instead, it would be secreted. There is evidence that endothelial cells make this alternatively spliced form and that it has angiogenic activity, perhaps explaining why it is found in the blood (Angiolini et al., 2019).
If L1CAM in the CSF and plasma is soluble, then why would neuronal markers come along when researchers fish out L1CAM with antibodies? Walt and colleagues had an answer for that, at least in the case of one marker. They report that the L1CAM antibody binds α-synuclein, as well.
That may be, but researchers have enriched for many other neuron markers by using anti-L1CAM antibodies for capture, including synaptophysin, tau, Aβ, neuregulin, neurogranin, and neurofilament light. “Do the authors suggest all these proteins nonspecifically interact with the L1CAM antibody?” asked Kapogiannis. “We have found that L1CAM EVs are enriched in neuronal-specific miRNAs, which have an even lower chance to nonspecifically interact with the antibody,” he wrote. Kapogiannis’ group also recently found that L1CAM associated with ALIX, a protein found in the lumen of extracellular vesicles (see full comment below).
Lynn Pulliam, University of California, San Francisco, told Alzforum that her group has tested the cargo of these L1CAM EVs extensively using Mass Spec, targeted ELISAs, and multiplex neurodegenerative protein panels. “The neuronal EVs we isolate contain abundant neuronal proteins,” she wrote. “Whether or not all these L1CAM EVs come from neurons is tough to say, since it is true L1CAM is found on a few non neuronal cells; however, we do believe the majority of the L1CAM isolated EVs are of neuronal origin.”
No consensus was reached at the ISEV/MJFF workshop on whether L1CAM identifies NDEVs, and there was no indication that researchers were going to quit using this protein to isolate vesicles any time soon. “I’m all open to using an alternative marker if we can find one,” said Kapogiannis. Some suggested neural cell adhesion molecule. “We have found that NCAM is cleaved also, so we don’t think that is good, either,” said Walt. Others challenged him to suggests a better marker. “We have looked at about half a dozen but have not found a good candidate,” he said.—Tom Fagan
References
Paper Citations
- Shi M, Liu C, Cook TJ, Bullock KM, Zhao Y, Ginghina C, Li Y, Aro P, Dator R, He C, Hipp MJ, Zabetian CP, Peskind ER, Hu SC, Quinn JF, Galasko DR, Banks WA, Zhang J. Plasma exosomal α-synuclein is likely CNS-derived and increased in Parkinson's disease. Acta Neuropathol. 2014 Nov;128(5):639-50. Epub 2014 Jul 6 PubMed.
- Goetzl EJ, Boxer A, Schwartz JB, Abner EL, Petersen RC, Miller BL, Kapogiannis D. Altered lysosomal proteins in neural-derived plasma exosomes in preclinical Alzheimer disease. Neurology. 2015 Jul 7;85(1):40-7. Epub 2015 Jun 10 PubMed.
- Winston CN, Goetzl EJ, Akers JC, Carter BS, Rockenstein EM, Galasko D, Masliah E, Rissman RA. Prediction of conversion from mild cognitive impairment to dementia with neuronally derived blood exosome protein profile. Alzheimers Dement (Amst). 2016;3:63-72. Epub 2016 May 7 PubMed.
- Pulliam L, Sun B, Mustapic M, Chawla S, Kapogiannis D. Plasma neuronal exosomes serve as biomarkers of cognitive impairment in HIV infection and Alzheimer's disease. J Neurovirol. 2019 Jan 4; PubMed.
- Cha DJ, Mengel D, Mustapic M, Liu W, Selkoe DJ, Kapogiannis D, Galasko D, Rissman RA, Bennett DA, Walsh DM. miR-212 and miR-132 Are Downregulated in Neurally Derived Plasma Exosomes of Alzheimer's Patients. Front Neurosci. 2019;13:1208. Epub 2019 Nov 26 PubMed.
- Jiang C, Hopfner F, Katsikoudi A, Hein R, Catli C, Evetts S, Huang Y, Wang H, Ryder JW, Kuhlenbaeumer G, Deuschl G, Padovani A, Berg D, Borroni B, Hu MT, Davis JJ, Tofaris GK. Serum neuronal exosomes predict and differentiate Parkinson's disease from atypical parkinsonism. J Neurol Neurosurg Psychiatry. 2020 Jul;91(7):720-729. Epub 2020 Apr 9 PubMed.
- Angiolini F, Belloni E, Giordano M, Campioni M, Forneris F, Paronetto MP, Lupia M, Brandas C, Pradella D, Di Matteo A, Giampietro C, Jodice G, Luise C, Bertalot G, Freddi S, Malinverno M, Irimia M, Moulton JD, Summerton J, Chiapparino A, Ghilardi C, Giavazzi R, Nyqvist D, Gabellini D, Dejana E, Cavallaro U, Ghigna C. A novel L1CAM isoform with angiogenic activity generated by NOVA2-mediated alternative splicing. Elife. 2019 Mar 4;8 PubMed.
External Citations
Further Reading
No Available Further Reading
Primary Papers
- Norman M, Ter-Ovanesyan D, Trieu W, Lazarovits R, Kowal EJ, Lee JH, Chen-Plotkin AS, Regev A, Church GM, Walt DR. L1CAM is not associated with extracellular vesicles in human cerebrospinal fluid or plasma. Nat Methods. 2021 Jun;18(6):631-634. Epub 2021 Jun 3 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
National Institute on Aging
The study by Norman et al. makes an important contribution to the ongoing research on deriving neuronal extracellular vesicles (EVs) from biofluids; however, I cannot accept its bold conclusions regarding L1CAM.
First, the study does not address (or even mention) the accumulated evidence on neuronal cargo enrichment from multiple studies of L1CAM immunoprecipitated EVs. The only attempt to address this topic is done by demonstrating that their L1CAM antibody captures recombinant α-synuclein to a greater degree (threefold) than does IgG. However, the authors’ interpretation of this finding is misleading, as 99 percent of α-synuclein in plasma is of non-neuronal origin.
Second, the study seeks to explain away the successful application of the methodology in deriving EV biomarkers for Alzheimer’s and many other brain conditions (including traumatic brain injury, frontotemporal dementia, Parkinson’s disease, multiple systems atrophy, restless legs syndrome, Neuro-l HIV, depression, schizophrenia, bipolar disorder), by using L1CAM to capture neuronal-derived EVs. These studies have shown enrichment for many neuronal proteins, such as synaptophysin, tau, neuronal enolase, proBDNF, neurofilament light, neuregulin, and many more. Do the authors suggest all these proteins nonspecifically interact with the L1CAM antibody? Moreover, we have found that L1CAM EVs are enriched in neuronal-specific miRNAs, which have an even lower chance to nonspecifically interact with the antibody.
Third, the authors’ main argument is that if no L1CAM is found in CSF and plasma fractions that contain tetraspanins, then this means that L1CAM in CSF is only the soluble form and L1CAM EVs do not exist (at least in these media). However, when they isolate EVs with size exclusion chromatography, they find high levels of L1CAM in late fractions that also contain CD81, CD63, and CD9. Since EVs come in a wide range of sizes (20-1000 nm), we believe (based on our own EM images) that the majority of L1CAM-positive NDEVs are in the lower size range and more likely to be contained in late SEC fractions.
Fourth, western blots presented in Fig 2c clearly demonstrate that the eluate of L1CAM pulldown from plasma contains both the internal and external domains of L1CAM, which is entirely consistent with the presence of whole-length L1CAM on EVs. It puzzles me that the authors do not offer this most parsimonious interpretation to their finding, but rather postulate, with little evidence, that plasma contains an alternatively spliced peptide consisting of the internal and external domains, but not the transmembrane domain.
Fifth, recently we produced high-resolution microscopy images clearly demonstrating the presence of L1CAM on the surface of Alix+ particles in the size range of EVs (see image above). This is in agreement with the authors’ own finding in their figure 1b that L1CAM is associated with EVs produced by neurons in culture.
In conclusion, I think that the current paper shows that soluble L1CAM exists in plasma and CSF, something already widely known and accepted, but does not show that L1CAM-associated EVs do not exist.
Mayo Clinic Florida
This brief communication paper from Norman and Ter-Ovnesyan et al. from David Walt’s laboratory and collaborators reports a carefully conducted study to determine whether L1CAM in the human CSF or plasma is enriched in EV and which isoforms are present in the biofluids.
The results are rather stunning, showing that most of the L1CAM is detected in the soluble fraction, with not much in EV fraction as determined by size exclusion chromatography and discontinuous sucrose gradient ultracentrifugation. They also found, using two epitope-specific L1CAM antibodies, that the majority of L1CAM in CSF is 200 kDa (presumably a cleaved form) whereas that in plasma is 220 kDa. Mass spectrometry of plasma-derived L1CAM didn’t detect the transmembrane domain of the molecule, which is encoded in Exon 25. They also report, using Genotype-Tissue Expression RNA-Seq data, that a significant fraction of L1CAM does not contain Exon 25, suggesting it is secreted as a soluble form. Finally, they show that an anti-L1CAM antibody reported in previous studies captures α-synuclein, arguing that this molecule could be enriched by using anti-L1CAM antibody for immunocapture of neuron-derived EVs in the biofluids.
The results are convincing and will shed new light on the re-evaluation of the L1CAM-based method for isolating neuron-derived EVs. Our lab also recently studied the protein composition of EVs isolated from iPSC-derived neurons and could not detect L1CAM by mass-spectrometry.
Although these new findings do not eliminate the possibility of the existence of L1CAM in EVs, which has been widely utilized in multiple publications, alternative neuron-specific molecules more enriched in EVs are warranted for the isolation of neuron derived EVs from biofluids.
With efforts to better understand brain function at a cellular level in vivo, but inability to safely perform brain biopsy in live individuals, some members of the scientific community have turned to the study of neuron-derived extracellular vesicles (NDEVs). To identify and separate NDEVs from heterogenous mixtures of cells, specific markers are used to guide separation techniques such as fractionation and immunocapture. Two such markers, NCAM1 and L1CAM, have emerged for the study of NDEVs (Fauré et al., 2006; Fiandaca et al., 2014; Shi et al., 2014; Sun et al., 2017). Recently it has been cautioned that these markers may not be sufficiently neuron-specific (Fowler et al., 2019). Now, a new study has put the reliability of L1CAM in further doubt.
In this brief communication Norman, Ter-Ovanesyan, and colleagues carefully assess the association of L1CAM with extracellular vesicles derived from both cerebrospinal fluid (CSF) and from plasma (Norman et al., 2021). Using biochemical, proteomic, and genetic data, they suggest that most L1CAM present in fractionated samples is not vesicle-associated. The authors also performed immunocapture experiments against L1CAM in both CSF and plasma. In a key finding, L1CAM pulldowns from plasma indicate the presence of full-length L1CAM, while pulldowns from CSF showed only the presence of truncated L1CAM lacking the C-terminal transmembrane domain. These results indicate that the extracellular vesicles immunocaptured from human samples using L1CAM are not neuron-derived.
Are previous findings with L1CAM cast in doubt by this new publication? While such findings may not necessarily be true for NDEVs, they may still be true with respect to L1CAM-positive extracellular vesicles from other tissues. New, complementary experiments will be required to extend the claims back to neurons or NDEVs. For example, it appears that L1CAM may still be NDEV-associated in rat brain (Dagur et al., 2020). It also remains to be demonstrated whether NCAM1 is also absent from NDEVs. In either case, a recently published set of guidelines from a large cohort of scientists studying extracellular vesicles offers suggestions for appropriately cautious methodology and interpretation of results (Théry et al., 2018).
References:
Fauré J, Lachenal G, Court M, Hirrlinger J, Chatellard-Causse C, Blot B, Grange J, Schoehn G, Goldberg Y, Boyer V, Kirchhoff F, Raposo G, Garin J, Sadoul R. Exosomes are released by cultured cortical neurones. Mol Cell Neurosci. 2006 Apr;31(4):642-8. Epub 2006 Jan 30 PubMed.
Fiandaca MS, Kapogiannis D, Mapstone M, Boxer A, Eitan E, Schwartz JB, Abner EL, Petersen RC, Federoff HJ, Miller BL, Goetzl EJ. Identification of preclinical Alzheimer's disease by a profile of pathogenic proteins in neurally derived blood exosomes: A case-control study. Alzheimers Dement. 2014 Aug 14; PubMed.
Shi M, Liu C, Cook TJ, Bullock KM, Zhao Y, Ginghina C, Li Y, Aro P, Dator R, He C, Hipp MJ, Zabetian CP, Peskind ER, Hu SC, Quinn JF, Galasko DR, Banks WA, Zhang J. Plasma exosomal α-synuclein is likely CNS-derived and increased in Parkinson's disease. Acta Neuropathol. 2014 Nov;128(5):639-50. Epub 2014 Jul 6 PubMed.
Sun B, Dalvi P, Abadjian L, Tang N, Pulliam L. Blood neuron-derived exosomes as biomarkers of cognitive impairment in HIV. AIDS. 2017 Sep 10;31(14):F9-F17. PubMed.
Fowler CD. NeuroEVs: Characterizing Extracellular Vesicles Generated in the Neural Domain. J Neurosci. 2019 Nov 20;39(47):9262-9268. PubMed.
Norman M, Ter-Ovanesyan D, Trieu W, Lazarovits R, Kowal EJ, Lee JH, Chen-Plotkin AS, Regev A, Church GM, Walt DR. L1CAM is not associated with extracellular vesicles in human cerebrospinal fluid or plasma. Nat Methods. 2021 Jun;18(6):631-634. Epub 2021 Jun 3 PubMed.
Dagur RS, Liao K, Sil S, Niu F, Sun Z, Lyubchenko YL, Peeples ES, Hu G, Buch S. Neuronal-derived extracellular vesicles are enriched in the brain and serum of HIV-1 transgenic rats. J Extracell Vesicles. 2020;9(1):1703249. Epub 2019 Dec 20 PubMed.
Théry C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R, Antoniou A, Arab T, Archer F, Atkin-Smith GK, Ayre DC, Bach JM, Bachurski D, Baharvand H, Balaj L, Baldacchino S, Bauer NN, Baxter AA, Bebawy M, Beckham C, Bedina Zavec A, Benmoussa A, Berardi AC, Bergese P, Bielska E, Blenkiron C, Bobis-Wozowicz S, Boilard E, Boireau W, Bongiovanni A, Borràs FE, Bosch S, Boulanger CM, Breakefield X, Breglio AM, Brennan MÁ, Brigstock DR, Brisson A, Broekman ML, Bromberg JF, Bryl-Górecka P, Buch S, Buck AH, Burger D, Busatto S, Buschmann D, Bussolati B, Buzás EI, Byrd JB, Camussi G, Carter DR, Caruso S, Chamley LW, Chang YT, Chen C, Chen S, Cheng L, Chin AR, Clayton A, Clerici SP, Cocks A, Cocucci E, Coffey RJ, Cordeiro-da-Silva A, Couch Y, Coumans FA, Coyle B, Crescitelli R, Criado MF, D'Souza-Schorey C, Das S, Datta Chaudhuri A, de Candia P, De Santana EF, De Wever O, Del Portillo HA, Demaret T, Deville S, Devitt A, Dhondt B, Di Vizio D, Dieterich LC, Dolo V, Dominguez Rubio AP, Dominici M, Dourado MR, Driedonks TA, Duarte FV, Duncan HM, Eichenberger RM, Ekström K, El Andaloussi S, Elie-Caille C, Erdbrügger U, Falcón-Pérez JM, Fatima F, Fish JE, Flores-Bellver M, Försönits A, Frelet-Barrand A, Fricke F, Fuhrmann G, Gabrielsson S, Gámez-Valero A, Gardiner C, Gärtner K, Gaudin R, Gho YS, Giebel B, Gilbert C, Gimona M, Giusti I, Goberdhan DC, Görgens A, Gorski SM, Greening DW, Gross JC, Gualerzi A, Gupta GN, Gustafson D, Handberg A, Haraszti RA, Harrison P, Hegyesi H, Hendrix A, Hill AF, Hochberg FH, Hoffmann KF, Holder B, Holthofer H, Hosseinkhani B, Hu G, Huang Y, Huber V, Hunt S, Ibrahim AG, Ikezu T, Inal JM, Isin M, Ivanova A, Jackson HK, Jacobsen S, Jay SM, Jayachandran M, Jenster G, Jiang L, Johnson SM, Jones JC, Jong A, Jovanovic-Talisman T, Jung S, Kalluri R, Kano SI, Kaur S, Kawamura Y, Keller ET, Khamari D, Khomyakova E, Khvorova A, Kierulf P, Kim KP, Kislinger T, Klingeborn M, Klinke DJ 2nd, Kornek M, Kosanović MM, Kovács ÁF, Krämer-Albers EM, Krasemann S, Krause M, Kurochkin IV, Kusuma GD, Kuypers S, Laitinen S, Langevin SM, Languino LR, Lannigan J, Lässer C, Laurent LC, Lavieu G, Lázaro-Ibáñez E, Le Lay S, Lee MS, Lee YX, Lemos DS, Lenassi M, Leszczynska A, Li IT, Liao K, Libregts SF, Ligeti E, Lim R, Lim SK, Linē A, Linnemannstöns K, Llorente A, Lombard CA, Lorenowicz MJ, Lörincz ÁM, Lötvall J, Lovett J, Lowry MC, Loyer X, Lu Q, Lukomska B, Lunavat TR, Maas SL, Malhi H, Marcilla A, Mariani J, Mariscal J, Martens-Uzunova ES, Martin-Jaular L, Martinez MC, Martins VR, Mathieu M, Mathivanan S, Maugeri M, McGinnis LK, McVey MJ, Meckes DG Jr, Meehan KL, Mertens I, Minciacchi VR, Möller A, Møller Jørgensen M, Morales-Kastresana A, Morhayim J, Mullier F, Muraca M, Musante L, Mussack V, Muth DC, Myburgh KH, Najrana T, Nawaz M, Nazarenko I, Nejsum P, Neri C, Neri T, Nieuwland R, Nimrichter L, Nolan JP, Nolte-'t Hoen EN, Noren Hooten N, O'Driscoll L, O'Grady T, O'Loghlen A, Ochiya T, Olivier M, Ortiz A, Ortiz LA, Osteikoetxea X, Østergaard O, Ostrowski M, Park J, Pegtel DM, Peinado H, Perut F, Pfaffl MW, Phinney DG, Pieters BC, Pink RC, Pisetsky DS, Pogge von Strandmann E, Polakovicova I, Poon IK, Powell BH, Prada I, Pulliam L, Quesenberry P, Radeghieri A, Raffai RL, Raimondo S, Rak J, Ramirez MI, Raposo G, Rayyan MS, Regev-Rudzki N, Ricklefs FL, Robbins PD, Roberts DD, Rodrigues SC, Rohde E, Rome S, Rouschop KM, Rughetti A, Russell AE, Saá P, Sahoo S, Salas-Huenuleo E, Sánchez C, Saugstad JA, Saul MJ, Schiffelers RM, Schneider R, Schøyen TH, Scott A, Shahaj E, Sharma S, Shatnyeva O, Shekari F, Shelke GV, Shetty AK, Shiba K, Siljander PR, Silva AM, Skowronek A, Snyder OL 2nd, Soares RP, Sódar BW, Soekmadji C, Sotillo J, Stahl PD, Stoorvogel W, Stott SL, Strasser EF, Swift S, Tahara H, Tewari M, Timms K, Tiwari S, Tixeira R, Tkach M, Toh WS, Tomasini R, Torrecilhas AC, Tosar JP, Toxavidis V, Urbanelli L, Vader P, van Balkom BW, van der Grein SG, Van Deun J, van Herwijnen MJ, Van Keuren-Jensen K, van Niel G, van Royen ME, van Wijnen AJ, Vasconcelos MH, Vechetti IJ Jr, Veit TD, Vella LJ, Velot É, Verweij FJ, Vestad B, Viñas JL, Visnovitz T, Vukman KV, Wahlgren J, Watson DC, Wauben MH, Weaver A, Webber JP, Weber V, Wehman AM, Weiss DJ, Welsh JA, Wendt S, Wheelock AM, Wiener Z, Witte L, Wolfram J, Xagorari A, Xander P, Xu J, Yan X, Yáñez-Mó M, Yin H, Yuana Y, Zappulli V, Zarubova J, Žėkas V, Zhang JY, Zhao Z, Zheng L, Zheutlin AR, Zickler AM, Zimmermann P, Zivkovic AM, Zocco D, Zuba-Surma EK. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7(1):1535750. Epub 2018 Nov 23 PubMed.
Oxford University
It is not yet known what percentage of L1CAM-positive extracellular vesicles is of neuronal origin and whether these vesicles are derived from the brain or peripheral nerves. Although the discovery of more specific neuronal extracellular vesicle makers would be beneficial in this respect, the value of L1CAM immunocapture for biomarker studies has been carefully accessed and established in numerous publications across cohorts and neurological diseases. Norman et al. found that L1CAM exists in a free form in plasma and CSF, something that was previously shown in cell-conditioned media, but they have not produced any rigorous data to disprove that a small fraction is present on extracellular vesicles as demonstrated by many other groups.
As extensively discussed in the recent ISEV/MJFF workshop on L1CAM, L1CAM-positive extracellular vesicles constitute less than 15 percent of total circulating extracellular vesicles, based on nanoparticle tracking analysis (Suppl. Fig. 4 in Jiang et al., 2020; Kapogiannis et al., 2015) and only a small fraction of this protein co-elutes with plasma extracellular vesicle fractions in size-exclusion chromatography (Stuendl et al., 2021).
The authors have completely dismissed the presence of the small amount of L1CAM in their size-exclusion chromatography (SEC) fractions enriched with other extracellular vesicle protein markers, including CD63, CD81, and CD9 (e.g. Fraction 10 in Fig. 1c for plasma and CSF). Fig. 1b clearly shows that the majority of L1CAM vesicles from cell-conditioned media eluted in this fraction and differently from recombinant L1CAM, which appears in later fractions. It is therefore likely that they have missed the relatively small number of L1CAM-positive extracellular vesicles in their preparations. This is further compounded by the fact that SEC and similar methods only recover 40-60 percent of extracellular vesicles at best (Boing et al., 2014; Lobb et al., 2015), and much lower yields have been reported (Baranyai et al., 2015).
To definitively disprove that there is no L1CAM on extracellular vesicles, the authors need to carefully examine fraction 10 in their SEC experiments, e.g., with immunogold labelling and electron microscopy.
As for nonspecific binding of α-synuclein to anti-L1CAM antibody-coated beads, instead of performing L1CAM immunocapture from plasma or CSF followed by SIMOA measurements, the authors chose to perform immunocapture in a solution with excess of recombinant α-synuclein, a condition that is prone to artefacts, irrespective of the antibody used, largely due to nonspecific binding of α-synuclein to certain types of beads, especially if they are not antifouling as we have recently emphasized (Fig 2. in Fu et al., 2020). Total plasma α-synuclein is not elevated in Parkinson’s disease (Shi et al., 2014), and therefore nonspecific binding of α-synuclein to anti-L1CAM antibodies cannot explain the detection of increased α-synuclein in L1CAM-positive extracellular vesicles isolated from patient samples.
It is telling that with IgG alone they immunocaptured more α-synuclein (~120 pg/ml) than what was previously determined to be the cut-off α-synuclein (~10-14 pg/ml) in immunocaptured L1CAM-positive extracellular vesicles for Parkinson’s disease diagnosis compared to controls, using patient serum/plasma across multiple different cohorts (Shi et al., 2014; Jiang et al., 2020; Jiang et al., 2021).
References:
Jiang C, Hopfner F, Katsikoudi A, Hein R, Catli C, Evetts S, Huang Y, Wang H, Ryder JW, Kuhlenbaeumer G, Deuschl G, Padovani A, Berg D, Borroni B, Hu MT, Davis JJ, Tofaris GK. Serum neuronal exosomes predict and differentiate Parkinson's disease from atypical parkinsonism. J Neurol Neurosurg Psychiatry. 2020 Jul;91(7):720-729. Epub 2020 Apr 9 PubMed.
Kapogiannis D, Boxer A, Schwartz JB, Abner EL, Biragyn A, Masharani U, Frassetto L, Petersen RC, Miller BL, Goetzl EJ. Dysfunctionally phosphorylated type 1 insulin receptor substrate in neural-derived blood exosomes of preclinical Alzheimer's disease. FASEB J. 2015 Feb;29(2):589-96. Epub 2014 Oct 23 PubMed.
Stuendl A, Kraus T, Chatterjee M, Zapke B, Sadowski B, Moebius W, Hobert MA, Deuschle C, Brockmann K, Maetzler W, Mollenhauer B, Schneider A. α-Synuclein in Plasma-Derived Extracellular Vesicles Is a Potential Biomarker of Parkinson's Disease. Mov Disord. 2021 May 18; PubMed.
Böing AN, van der Pol E, Grootemaat AE, Coumans FA, Sturk A, Nieuwland R. Single-step isolation of extracellular vesicles by size-exclusion chromatography. J Extracell Vesicles. 2014;3 Epub 2014 Sep 8 PubMed.
Lobb RJ, Becker M, Wen SW, Wong CS, Wiegmans AP, Leimgruber A, Möller A. Optimized exosome isolation protocol for cell culture supernatant and human plasma. J Extracell Vesicles. 2015;4:27031. Epub 2015 Jul 17 PubMed.
Baranyai T, Herczeg K, Onódi Z, Voszka I, Módos K, Marton N, Nagy G, Mäger I, Wood MJ, El Andaloussi S, Pálinkás Z, Kumar V, Nagy P, Kittel Á, Buzás EI, Ferdinandy P, Giricz Z. Isolation of Exosomes from Blood Plasma: Qualitative and Quantitative Comparison of Ultracentrifugation and Size Exclusion Chromatography Methods. PLoS One. 2015;10(12):e0145686. Epub 2015 Dec 21 PubMed.
Fu Y, Jiang C, Tofaris GK, Davis JJ. Facile Impedimetric Analysis of Neuronal Exosome Markers in Parkinson's Disease Diagnostics. Anal Chem. 2020 Oct 20;92(20):13647-13651. Epub 2020 Oct 5 PubMed.
Shi M, Liu C, Cook TJ, Bullock KM, Zhao Y, Ginghina C, Li Y, Aro P, Dator R, He C, Hipp MJ, Zabetian CP, Peskind ER, Hu SC, Quinn JF, Galasko DR, Banks WA, Zhang J. Plasma exosomal α-synuclein is likely CNS-derived and increased in Parkinson's disease. Acta Neuropathol. 2014 Nov;128(5):639-50. Epub 2014 Jul 6 PubMed.
Jiang C, Hopfner F, Berg D, Hu MT, Pilotto A, Borroni B, Davis JJ, Tofaris GK. Validation of α-Synuclein in L1CAM-Immunocaptured Exosomes as a Biomarker for the Stratification of Parkinsonian Syndromes. Mov Disord. 2021 Apr 7; PubMed.
University of Goteborg, Sahlgrenska University Hospital
This paper by Norman and colleagues on the use of the L1CAM protein to enrich for “neuron-derived extracellular vesicles” (NDVEs), also called neuronally derived “exosomes,” is an important contribution on methodological aspects in the search for blood biomarkers for Alzheimer’s disease (AD).
Blood biomarkers have long been sought as a simple and easily accessible substitute for cerebrospinal fluid (CSF) or positron-emission tomography AD biomarkers. However, while AD biomarkers such as phosphorylated tau (p-tau181) can be measured in CSF using ELISA methods (Vanmechelen et al., 2000), the very low abundance of p-tau in blood (plasma or serum) precluded quantification (and detection) of this biomarker in blood samples using standard immunoassays. Only recently, the development of ultrasensitive immunoassay technology paved the way for the development of methods to accurately quantify p-tau181 in plasma and serum (see, e.g., Janelidze et al., 2020; Karikari et al., 2020).
However, some years ago, a series of papers proposed that p-tau181 and other AD biomarkers could be measured using standard ELISA methods after an enrichment procedure for neuronally derived “exosomes” from serum or plasma samples (see e.g., Fiandaca et al., 2014), and neuronally derived exosomes in plasma have been proposed as useful tools for understanding AD pathophysiology and for drug development (Goetzl, 2020). Many of these papers used the ExoQuick exosome precipitation kit, in which exosomes are precipitated from 0.5 mL of plasma or serum, and “neuronally-derived” exosomes are immunocaptured using a biotinylated L1CAM antibody, and after acid extraction finally resuspended to the same volume (0.5 mL) as the starting volume for plasma or serum (Fiandaca et al., 2014). Levels of p-tau181 (and other AD biomarkers) were remarkably high, even higher than in CSF.
Norman and co-workers elegantly show that the L1CAM protein is not associated with extracellular vesicles in human plasma or CSF samples, but is instead present as a soluble protein (Norman et al., 2021). This means that in protocols (or kits) using an L1CAM antibody for immunoprecipitation, the soluble protein (instead of neuronally derived exosomes) are captured, and therefore they recommend against the use of L1CAM in NDEV protocols. Without doubt, this paper shows the importance of proper analytical validation of analytical tools for biomarker measurements.
We have in my laboratory put efforts into evaluating the ExoQuick exosome precipitation kit, but while measured levels of p-tau181 (and other AD biomarkers) were high in the extracts, we were unable to find any differences in biomarker levels between AD and control cases. Instead, we noted that there also was a biotin-interference in ELISA methods based on biotinylated antibodies (such as the Innotest p-tau ELISA) that gave a marked unspecific ELISA signal (and measured “p-tau” levels), probably from contamination by the biotinylated L1CAM antibody (or leakage of biotin) when using the ExoQuick kit to isolate exosomes (unpublished data).
Nevertheless, the question remains whether any differences in biomarker levels between AD patients and controls can be replicated using other protocols, that are analytically validated, for isolation of peripheral blood exosomes.
References:
Vanmechelen E, Vanderstichele H, Davidsson P, Van Kerschaver E, Van Der Perre B, Sjögren M, Andreasen N, Blennow K. Quantification of tau phosphorylated at threonine 181 in human cerebrospinal fluid: a sandwich ELISA with a synthetic phosphopeptide for standardization. Neurosci Lett. 2000 May 5;285(1):49-52. PubMed.
Janelidze S, Mattsson N, Palmqvist S, Smith R, Beach TG, Serrano GE, Chai X, Proctor NK, Eichenlaub U, Zetterberg H, Blennow K, Reiman EM, Stomrud E, Dage JL, Hansson O. Plasma P-tau181 in Alzheimer's disease: relationship to other biomarkers, differential diagnosis, neuropathology and longitudinal progression to Alzheimer's dementia. Nat Med. 2020 Mar;26(3):379-386. Epub 2020 Mar 2 PubMed.
Karikari TK, Pascoal TA, Ashton NJ, Janelidze S, Benedet AL, Rodriguez JL, Chamoun M, Savard M, Kang MS, Therriault J, Schöll M, Massarweh G, Soucy JP, Höglund K, Brinkmalm G, Mattsson N, Palmqvist S, Gauthier S, Stomrud E, Zetterberg H, Hansson O, Rosa-Neto P, Blennow K. Blood phosphorylated tau 181 as a biomarker for Alzheimer's disease: a diagnostic performance and prediction modelling study using data from four prospective cohorts. Lancet Neurol. 2020 May;19(5):422-433. PubMed.
Fiandaca MS, Kapogiannis D, Mapstone M, Boxer A, Eitan E, Schwartz JB, Abner EL, Petersen RC, Federoff HJ, Miller BL, Goetzl EJ. Identification of preclinical Alzheimer's disease by a profile of pathogenic proteins in neurally derived blood exosomes: A case-control study. Alzheimers Dement. 2014 Aug 14; PubMed.
Goetzl EJ. Advancing medicine for Alzheimer's disease: A plasma neural exosome platform. FASEB J. 2020 Oct;34(10):13079-13084. Epub 2020 Aug 28 PubMed.
Norman M, Ter-Ovanesyan D, Trieu W, Lazarovits R, Kowal EJ, Lee JH, Chen-Plotkin AS, Regev A, Church GM, Walt DR. L1CAM is not associated with extracellular vesicles in human cerebrospinal fluid or plasma. Nat Methods. 2021 Jun;18(6):631-634. Epub 2021 Jun 3 PubMed.
NeuroDex INC
We at NeuroDex find the study by Norman et al. to be very interesting because it sheds more light on the high heterogeneity of extracellular vesicles (EVs) in terms of size and content. While we think that the study results are mostly solid, we do not agree with their interpretation.
In the size-exclusion chromatography (SEC) experiment the authors detect low levels of L1CAM in the early fraction and low levels of tetraspanins in later fractions. This overlap between L1CAM and tetraspanins suggests that L1CAM may be on a small fraction of EVs, it is most probable that these are smaller EVs. For example, fraction 9 in their SEC experiment (figure 1C) contains high levels of CD81 and CD63, and at least 100 pg/ml of L1CAM. The electron microscopy (EM) image they provided also showed that the fraction contains large EVs. Importantly, the EM image of fraction 12, which contains the highest level of L1CAM, is of poor quality and low amplification, which makes it hard to determine if small EVs (which can be as small as 20 nM) are present. This is in agreement with some of our own studies and with Dr. Walt’s recent biorRXiv paper (Ter-Ovanesyan et al., 2021) showing that intact EV SIMOA assay detects signals in late SEC fractions.
Besides commenting directly on this report, we would like to mention that while we see L1CAM associated with plasma-neuron-derived EVs, we do not think it is the optimal marker for their enrichment. L1CAM is not the most specific marker, and its existence as a soluble protein interferes with the immunoaffinity isolation and reduces the enrichment efficiency. Therefore, NeuroDex has scanned a large number of neuronal-specific surface markers (more than 60 different antibodies) and we identified markers that provide better enrichment than L1CAM, when neuronal proteins and RNA are measured. Using intact EV analysis, we have shown that few of the neuronal specific markers co-localize on the same EV, but they are not co-localized with nonspecific markers such as CD235 (erythrocyte marker). Moreover, the enrichment of neuronal cargo after neruonal EV isolation with our methodology has been demonstrated using unbiased approaches such as proteomics and RNA-seq analysis.
It is important to note that immunoaffinity methodology provides enrichment and not purification, for example we see about 20-fold increase in the neuronal to non-neuronal signals in the neuron-derived exosome fraction compared to neat plasma and/or total EVs.
Finally, we agree with Dr. Witwer that the biology and diagnosis potential are two separate issues. L1CAM-derived EVs have been shown to have diagnostic potential by several independent groups, with well over 2,000 samples analyzed. Such vast clinical data shouldn’t be ignored because of this report. It also generates an entry barrier for new enrichment markers like ours, and we as well as others have the obligation to not only prove the specificity of markers for neuron derived exosomes, but also that their clinical potential is better than for enrichment with L1CAM.
References:
Ter-Ovanesyan D, Norman M, Lazarovits R, Trieu W, Lee JH, Church GM, Walt DR. Framework for rapid comparison of extracellular vesicle isolation methods. Elife. 2021 Nov 16;10 PubMed.
University of Washington
University of Washington
The First Affiliated Hospital, Zhejiang University School of Medicine
The recent publication by Norman et al. suggests that L1CAM is not associated with extracellular vesicles (EVs) in human cerebrospinal fluid (CSF) or plasma; however, the conclusion contrasts sharply with a large body of evidence that already exists in the literature and is discussed in much greater detail in following sections.
1. Is L1CAM present on EVs isolated from blood (plasma) and other body fluids?
In 2014, we initially proposed that L1CAM, an EV surface protein that is highly expressed in neurons, could be used to enrich likely central nervous system (CNS)- or neuron-derived EVs (NDEs) in peripheral blood (plasma), and that α-synuclein contained in these plasma NDEs was diagnostically and prognostically relevant to Parkinson disease (Shi et al., 2014). Since then, L1CAM has been widely used to enrich NDEs from blood and other body fluids by independent groups, and these NDEs, as well as the carried proteins and RNAs, have shown substantial potential as “windows into the brain,” points of transparency allowing examination of peripheral body fluids to reveal CNS disorder-associated changes in brain biochemistry and intercellular communication (see Shi et al., 2019, for a recent review).
Norman et al. used size-exclusion chromatography (SEC) and density gradient centrifugation to separate EVs from soluble proteins and found that L1CAM was primarily detected in the soluble protein (e.g., albumin) fractions (a result that is similar to our unpublished data), but not in the EV protein (e.g., tetraspanins) fractions. They concluded that “L1CAM is not associated with extracellular vesicles in human plasma or cerebrospinal fluid.” The latter conclusion is, unfortunately, associated with many caveats that were not addressed.
(a) The presence of free L1CAM does not disprove the additional presence of EV-associated L1CAM (expected to be of low abundance in blood when derived from neurons or CNS); indeed, even in the Norman study an overlap between L1CAM and tetraspanin (general EV surface proteins) fractions was observed (Figures 1 and 2). Further, support for L1CAM’s association with EVs in plasma and other body fluids can be found in numerous investigations with multiple platforms, including nanoparticle tracking analysis (Shi et al., 2016; Winston et al., 2016; Mustapic et al., 2017; Kapogiannis et al., 2019), electron microscopy (EM), and immuno-EM (Shi et al., 2014; Mustapic et al., 2017; Kapogiannis et al., 2019; Jia et al., 2019).
In the past one to two years, several independent groups also presented evidence to support the presence of L1CAM in EV fractions isolated by SEC and other technologies in various scientific meetings, including the recent ISEV/MJFF-sponsored symposium. For example, Kapogiannis’s group presented evidence of L1CAM on EVs using cryo-EM and flow-based technologies. We have recently also acquired additional supportive evidence by using stochastic optical reconstruction microscopy (STORM) and Apogee nanoscale flow cytometry-based technologies (unpublished data). Together, these data suggest that, while Norman et al. may well be correct in that the predominant form of L1CAM in CSF and plasma is the free form, the presented evidence does not disprove the significant data supporting that some fraction of L1CAM is clearly associated with EVs.
(b) To measure the levels of L1CAM and other proteins in EV and non-EV fractions from plasma and CSF, Norman et al. used SIMOA assays developed in their lab. Although SIMOA is a highly sensitive assay platform, like any other immunoassays, SIMOA, particularly the “homemade” ones, need to be rigorously tested for their performance in the target sample matrix. The Norman study provided convincing data regarding the assay accuracy from dilution linearity and spiking recovery experiments (Supplementary Figure 1 and Supplementary Table 1). Nevertheless, a few other critical pieces of information, including assay sensitivity (Lower Limits of Detection, or LLOD, were provided in Supplementary Figure 2 for tetraspanins but not for L1CAM; Lower Limits of Quantification, or LLOQ, were not provided), specificity, and reproducibility were missing. Most investigators would agree that failure to detect a protein usually suggests the protein levels might be below the detection limits of the assays, but cannot be used to prove that the protein does not exist. In the same line of argument, as acknowledged by the authors: Absence of membrane-bound L1CAM in the proteomics analysis is inconclusive, as it is well-known in the field that absence of a protein in a mass spec analysis does not mean that it is not there.
(c) Loss of L1CAM in their study should be considered. As indicated in the discussions during the recent ISEV/MJFF-sponsored meeting, identification of L1CAM on EVs using the methods described requires that the association survives the preparation process. Careful sample processing, e.g., inclusion of appropriate protease inhibitors, and maintaining low temperature throughout the process, is thus critical for optimal results. Without these key components in the procedure, it is entirely possible that the sub-population of EVs carrying L1CAM, which most groups agree is a small proportion of the total EV fraction, especially in plasma, may be further diminished, either by loss of material, or, as the authors themselves suggest, by the cleavage of L1CAM extracellular domains, releasing the protein from the EVs. In particular, their mass spectrometry and other results appear to be in line with the conclusion that the majority of L1CAM in their preparations might be cleavage (rather than alternative splicing) products. Again, detection of the protein within each fraction is dependent on the sensitivity of the assay.
In short, several independent groups have reported the detection of L1CAM in EV fractions, and a few pieces of evidence are unequivocal. A lack of L1CAM detection likely reflects a less-optimal enrichment of the starting material, and/or insufficient sensitivity for the (remaining) protein, rather than its absence in the starting material.
2. Is L1CAM specific to CNS or neurons?
As recognized by many groups, in addition to neurons and other brain cells, L1CAM is also known to be expressed in certain cancer and specialized cells, including some kidney cells (Kenwrick et al., 2000). However, in addition to the reported relevance of cargos in L1CAM-positive EVs to CNS disorders, published and unpublished proteomic studies demonstrated that L1CAM-containing EVs in plasma have higher concentrations of several markers (e.g., phosphorylated tau, neuron-specific enolase, microtubule-associated protein 2, neurofilament light chain, and L1CAM) that are reasonably specific to neurons than do total plasma EVs, highly suggesting a neuronal (and, to some degree, CNS) origin of these EVs (Mustapic et al., 2017) or at least the enrichment of CNS- or neuron-derived EVs (Mustapic et al., 2017). Furthermore, there is currently no evidence indicating that L1CAM-containing EVs are secreted from kidney and other peripheral cell types.
Therefore, while we do agree that the CNS- or neuron- specificity of L1CAM-containing EVs in blood and the associated biological processes need to be further investigated, and that more CNS- or neuron-specific EV markers could be further pursued, L1CAM should not be discouraged in this line of investigations simply because of a study that is associated with many caveats.
3. Is α-synuclein detected in L1CAM-enriched EVs due to non-specific binding of α-synuclein to anti-L1CAM antibodies?
The authors argue that measurements of α-synuclein, purported to be vesicle-contained, are confounded by the interaction of α-synuclein with the anti-L1CAM antibody, but not as much with the IgG isotype control. While this point has its merits (as demonstrated in Extended Data Figure 6), the lack of a detailed description of their immunoprecipitation (IP) procedure and reagents used in the study prevents verification of these results by others. Additionally, the amounts of recombinant α-synuclein used in the IP experiment appeared to be much lower than those in plasma (usually in tens of ng/mL), suggesting that the phenomenon, even true, may not be relevant in real human samples.
Furthermore, while most researchers would not argue that α-synuclein readily associates with many antibodies and target proteins, independent groups have presented evidence that conflicts with the conclusions presented in this Nat Methods article. For example, the α-synuclein signal detected in EVs isolated by immunocapture was nearly eliminated if the sample was pretreated by ultracentrifugation (Shi et al., 2014). While it can be argued that ultracentrifugation could reduce α-synuclein in the sample, particularly large aggregates, plasma contains a very high concentration of the protein, and it would be expected that if the primary source of α-synuclein in the final processed sample was nonspecific binding to the anti-L1CAM antibodies, this should still be a significant contributor to the signal. Moreover, a large number of other targets have also been measured using the L1CAM-targeting EV isolation strategy, including Aβ, tau, lysosomal proteins, synaptic proteins, and others (see (Shi et al., 2019, for a recent review). It is highly unlikely that nonspecific binding explains the presence of all of these target putative cargoes across studies.
More importantly, even if true, higher nonspecific affinity of anti-L1CAM for α-synuclein cannot explain the observed disease (e.g., PD) and control group differences for α-synuclein and other proteins, which has been confirmed by independent groups in multiple studies (Zhao et al., 2018; Niu et al., 2020; Jiang et al., 2021).
In summary, while Norman et al.’s study certainly highlights important caveats associated with L1CAM investigations, which should be considered in future studies focused on L1CAM-based identification of EVs, we believe further investigations on the biology and biomarker usages of L1CAM-EVs should not be discouraged.
References:
Shi M, Liu C, Cook TJ, Bullock KM, Zhao Y, Ginghina C, Li Y, Aro P, Dator R, He C, Hipp MJ, Zabetian CP, Peskind ER, Hu SC, Quinn JF, Galasko DR, Banks WA, Zhang J. Plasma exosomal α-synuclein is likely CNS-derived and increased in Parkinson's disease. Acta Neuropathol. 2014 Nov;128(5):639-50. Epub 2014 Jul 6 PubMed.
Shi M, Sheng L, Stewart T, Zabetian CP, Zhang J. New windows into the brain: Central nervous system-derived extracellular vesicles in blood. Prog Neurobiol. 2019 Apr;175:96-106. Epub 2019 Jan 25 PubMed.
Shi M, Kovac A, Korff A, Cook TJ, Ginghina C, Bullock KM, Yang L, Stewart T, Zheng D, Aro P, Atik A, Kerr KF, Zabetian CP, Peskind ER, Hu SC, Quinn JF, Galasko DR, Montine TJ, Banks WA, Zhang J. CNS tau efflux via exosomes is likely increased in Parkinson's disease but not in Alzheimer's disease. Alzheimers Dement. 2016 Nov;12(11):1125-1131. Epub 2016 May 24 PubMed.
Winston CN, Goetzl EJ, Akers JC, Carter BS, Rockenstein EM, Galasko D, Masliah E, Rissman RA. Prediction of conversion from mild cognitive impairment to dementia with neuronally derived blood exosome protein profile. Alzheimers Dement (Amst). 2016;3:63-72. Epub 2016 May 7 PubMed.
Mustapic M, Eitan E, Werner JK Jr, Berkowitz ST, Lazaropoulos MP, Tran J, Goetzl EJ, Kapogiannis D. Plasma Extracellular Vesicles Enriched for Neuronal Origin: A Potential Window into Brain Pathologic Processes. Front Neurosci. 2017;11:278. Epub 2017 May 22 PubMed.
Kapogiannis D, Mustapic M, Shardell MD, Berkowitz ST, Diehl TC, Spangler RD, Tran J, Lazaropoulos MP, Chawla S, Gulyani S, Eitan E, An Y, Huang CW, Oh ES, Lyketsos CG, Resnick SM, Goetzl EJ, Ferrucci L. Association of Extracellular Vesicle Biomarkers With Alzheimer Disease in the Baltimore Longitudinal Study of Aging. JAMA Neurol. 2019 Jul 15; PubMed.
Jia L, Qiu Q, Zhang H, Chu L, Du Y, Zhang J, Zhou C, Liang F, Shi S, Wang S, Qin W, Wang Q, Li F, Wang Q, Li Y, Shen L, Wei Y, Jia J. Concordance between the assessment of Aβ42, T-tau, and P-T181-tau in peripheral blood neuronal-derived exosomes and cerebrospinal fluid. Alzheimers Dement. 2019 Aug;15(8):1071-1080. PubMed.
Kenwrick S, Watkins A, De Angelis E. Neural cell recognition molecule L1: relating biological complexity to human disease mutations. Hum Mol Genet. 2000 Apr 12;9(6):879-86. PubMed.
Zhao ZH, Chen ZT, Zhou RL, Zhang X, Ye QY, Wang YZ. Increased DJ-1 and α-Synuclein in Plasma Neural-Derived Exosomes as Potential Markers for Parkinson's Disease. Front Aging Neurosci. 2018;10:438. Epub 2019 Jan 14 PubMed.
Niu M, Li Y, Li G, Zhou L, Luo N, Yao M, Kang W, Liu J. A longitudinal study on α-synuclein in plasma neuronal exosomes as a biomarker for Parkinson's disease development and progression. Eur J Neurol. 2020 Jun;27(6):967-974. Epub 2020 Apr 13 PubMed.
Jiang C, Hopfner F, Berg D, Hu MT, Pilotto A, Borroni B, Davis JJ, Tofaris GK. Validation of α-Synuclein in L1CAM-Immunocaptured Exosomes as a Biomarker for the Stratification of Parkinsonian Syndromes. Mov Disord. 2021 Apr 7; PubMed.
IIBB/CSIC/CIBERNED
The study by Norman et al. concludes that L1CAM is not associated with extracellular vesicles (EVs) in human plasma or cerebrospinal fluid. The study assumes that all EVs contain tretraspanins, like classical exosomes. However, this assumption is not supported by evidence, because it excludes other types of EVs (non-classical exosomes, microvesicles, or autophagic EVs).
The study clearly shows that there is soluble L1CAM in CSF and plasma, but does not explore whether in these biofluids there are L1CAM-positive EVs that do not display tetraspanins. The authors clearly show the presence of L1CAM-positive EVs in the conditioned medium of human-induced pluripotent cell-derived neurons (iNGNs). However, one piece of critical information that would support, or not, their conclusion is whether these L1CAM-positive EVs from iNGNs do, or do not, display tetraspanins.
Sant Pau Biomedical Research Institute, Memory Unit
Sant Pau Biomedical Research Institute, Memory Unit
St Paul Biomedical Research Institute
Make a Comment
To make a comment you must login or register.