Liquid Phase Transition: A Deluge of Data Points to Multiple Regulators
Quick Links
For better or worse, liquid-liquid phase separation (LLPS) happens. It’s beneficial when RNA and RNA-binding proteins (RBPs) form dynamic liquid assemblies required for many cell functions. It’s pathological when the same assemblies yield the toxic fibrils that mark many neurodegenerative diseases. In a leap forward, six papers illuminate new pathways that promote the better, and prevent the worse. It’s all about protein chaperones, posttranslational modifications, and RNA structures.
- Nuclear-import receptors and arginine methylation regulate phase separation and aggregation of RNA-binding proteins in the cytosol.
- These receptors dissolve fibrils and hydrogels, and mitigate neurodegeneration in model systems.
- In the nucleus, high RNA concentrations prevent aggregation of FUS, other RBPs.
Four papers in the April 19 Cell reveal that nuclear import receptors (NIRs) potently inhibit and even reverse phase transition and fibril formation of several pathogenic RBPs. Two studies, one from the labs of Yuh Min Chook and Michael Rosen, University of Texas Southwestern Medical Center, Dallas, and another led by James Shorter at the University of Pennsylvania in Philadelphia and Paul Taylor, St. Jude’s Children’s Research Hospital, Memphis, Tennessee, show that transportin proteins, best known for shuttling RBPs into the nucleus, appear to have another function. In the cytosol, they protect against aberrant aggregation of disease-related RBPs, including FUS, TDP-43, and others. Alzforum first reported on this data from the Phase Transitions in Biology and Disease meeting in Leuven last May (May 2017 conference news). Independently, Peter St George-Hyslop of the University of Toronto and Dorothee Dormann at Ludwig-Maximilians-University in Munich uncovered that transportin tames FUS. They also report that arginine methylation inhibits phase transition of FUS, and that these pathways go awry in some cases of amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD).
“All these papers converge on the same findings, showing two new pathways that regulate aggregation,” said Ben Wolozin of Boston University. “They establish that nuclear import proteins also function as disaggregases, acting like the Jaws of Life to untangle protein wreckage, and that methylation at least partly regulates that process. That gives us an extra target to try to interfere with these processes,” he told Alzforum.
All this occurs in the cytosol, but what about in the nucleus, where RPBs concentrate and cytosolic chaperones are taboo? What keeps FUS and other RPBs from undergoing LLPS or aggregating there? In the April 12 Science, Simon Alberti, Anthony Hyman, and colleagues at the Max Planck Institute, Dresden, Germany, report that high concentrations of RNA do the trick. And finally, to add texture to it all, researchers led by Amy Gladfelter, University of North Carolina, Chapel Hill, report in the same issue that the shape of RNA determines the content, biophysical properties, and phase separation of RNA-protein assemblies, proving that there is much more to know about these particles and their regulation in health and disease.
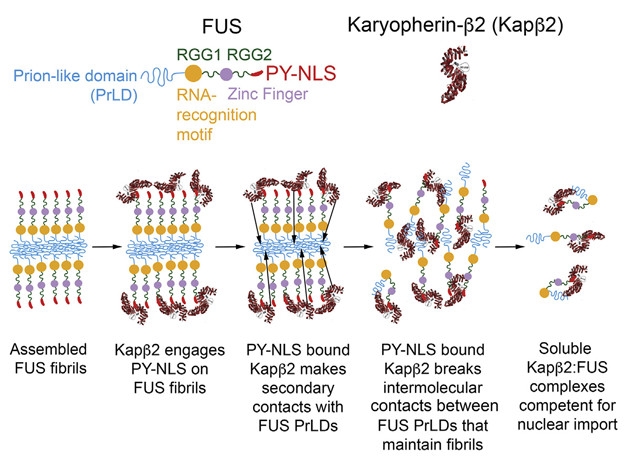
Fibrils Out. Kapβ2, aka TNPO1, binds to the PY-NLS of FUS, dissolves fibrils by interfering with weak FUS-FUS interactions, and ushers soluble FUS monomers back to the nucleus. [Courtesy of Guo et al., Cell 2018.]
Cytosolic Chaperones
The connection of nuclear import receptors to LLPS casts new light on an old relationship. Chook has long studied NIR interactions with FUS (Zhang and Chook, 2012). One of them, transportin-1 (TNPO1, aka Karyopherin-β2 or Kapβ2) recognizes a proline- and tyrosine-containing nuclear localization sequence (PY-NLS) on the C-terminus of FUS and on other RNA-binding proteins. Once bound to this sequence, TNPO1 directs translocation of proteins into the nucleus. Some disease-causing FUS mutations disrupt nuclear-cytoplasmic shuttling, leading to mislocalization and aggregation of the protein in the cytosol. But could there be more to the story? Chook and Shorter wondered if a tightly bound NIR protein might affect the ability of proteins like FUS to undergo phase transition in the cytoplasm. Rosen was already studying the physiological role of phase transition (Li et al., 2012), and they teamed up to tackle the problem. To start, co-first authors Takuya Yoshizawa in the Chook lab and Rustam Ali in the Rosen lab did a simple experiment: They added TNPO1 to purified FUS. The result was clear: TNPO1 potently prevented LLPS, and could even dissolve droplets of already liquefied FUS (see movie). This required an intact NLS, they showed.
Fading FUS. Liquid droplets formed from purified FUS protein dissipate within five minutes of TNPO1 addition. [Courtesy of Yoshizawa et al., Cell 2018.]
From there, the investigators probed the interaction of the FUS and TNPO1. After TNPO1 bound tightly to the FUS NLS, it made multiple weak interactions with other parts of the FUS protein. TNPO1 contacted multiple tyrosine-containing motifs in the N-terminal low-complexity prion-like domain, including the SYSGY sequence that forms reversible, amyloid-like structures, and the solid core of FUS fibers (Feb 2018 news; Apr 2018 news; Sep 2017 news). The protein interacted with portions of the FUS C-terminal RNA-binding and arginine-rich domains, which the investigators showed also contribute to LLPS. Though weak, collectively these multivalent interactions appear sufficient to disrupt FUS-FUS assembly, and prevent or reverse LLPS.
Shorter and Taylor’s study took the work in a different direction. First authors Lin Guo and Hejia Wang in Philadelphia and Hong Joo Kim in Memphis expanded the range of TNPO1, demonstrating the protein could prevent not just LLPS, but also fibrillization of purified FUS. TNPO1’s actions extended to other RBPs with a PY-NLS, including TAF15, EWSR1, hnRNPA1, and hnRNPA2. The exception was TDP-43, which bears a different NLS and was not affected by TNPO1. However, when researchers swapped in the appropriate transportin, a complex of Importin-α plus karyopherin β1, it successfully prevented aggregation of TDP-43.
Even more, TNPO1 proved to be a potent disaggregase, rapidly dissolving preformed fibrils and fibril-containing hydrogels formed by FUS and other RBPs, in some cases within minutes. That was surprising, Shorter told Alzforum. He had previously identified prion disaggregases in yeast, but none had been found in mammalian cells. “We really didn’t expect that it would reverse aggregation, but we could even see it on a macroscopic level. When we constructed hydrogels and dropped the protein on top, it melted those gels,” he said.
What about mutated, pathogenic forms of FUS? Mutations in the prion-like low-complexity domain often accelerate aberrant phase transition. Those mutants, and many in the NLS, were still susceptible to disaggregation by TNPO1. However, the protein could not solubilize FUS with NLS mutations that severely reduced TNPO1-NLS interactions. In particular, two FUS NLS mutations that destroyed TNPO1 binding partially or entirely resisted disaggregase activity. Interestingly, those two mutants, R495X and P525L, both cause an early, severe form of ALS.
Moving to in vivo studies, when the investigators boosted expression of TNPO1 in yeast, they were able to inhibit RBP aggregation. In mammalian cells, TNPO1 overexpression abrogated accumulation of mutant FUS in stress granules, and restored FUS expression in the nucleus, while preventing its toxicity. In fibroblasts from ALS patients with an R521H FUS mutation, increased expression of TNPO1 reversed a FUS loss-of-function phenotype, namely reduction in specific mRNAs. In flies, TNPO1 lessened neurodegeneration due to mutated FUS or mutated hnRNAP2, another RBP linked to FTLD.
The next step, Shorter said, will be to assess transportins in an animal model, to see if the proteins are protective there, too. “We predict they would be, but we have to do it,” he said.
Methyl Modifications
In addition to nuclear import receptors, posttranslational modification of FUS plays a critical role in phase separation, say two additional papers. The first, from the Dormann lab, builds on her previous work on FUS in ALS and FTLD. She had determined that in ALS-FUS, mutations affecting the NLS disrupt nuclear transport mediated by TNPO1, and thus cellular localization (Jul 2010 news). In FTLD-FUS patients who have no FUS mutations, she found that FUS arginine methylation was reduced, which also disrupted nuclear transport (Sep 2012 news).

Protein Problems.
New protein synthesis (heat map) in axon tips from Xenopus retinal neurons decreases when FUS is hypomethylated (right) compared with normal protein (left). Expression of TNPO1 corrects the defect (not shown). [Courtesy of Qamar et al., Cell 2018.]
In the new study, first authors Mario Hofweber and Saskia Hutten found that either loss of TNPO binding or arginine hypomethylation of C-terminal arginine-rich motifs enhanced phase separation of FUS in vitro and in vivo. The results unify the two disease pathways, Dormann told Alzforum, in that both NLS mutation and hypomethylation lead to increased phase separation, then aberrant FUS accumulation in stress granules.
The fourth and final Cell paper extends that work. St George-Hyslop and colleagues dove deep into the biophysical consequences of FUS hypomethylation and how it enhances phase separation of the protein. In the study, seven first authors—Seema Qamar, GuoZhen Wang, Suzanne Randle, Francesco Simone Ruggeri, Juan Varela, Julie Qiaojin Lin, and Emma Phillips—cover the experimental spectrum from physics to biology. Using a series of physical tools, they establish that in hypomethylated FUS, cation-π bonds between arginine residues in the C-terminal domain and tyrosines in the low-complexity domain likely initiate phase separation, which is then followed by stabilizing intermolecular β-sheet hydrogen bonding.
Those changes appear to contribute to FUS pathogenicity by disrupting protein synthesis. Regulated, local protein synthesis is required for synaptic function and plasticity. The weakening of this activity is a common denominator between ALS-FUS and FTLD-FUS, and likely underpins the cause of neuronal injury in these two FUS-related diseases, St George-Hyslop told Alzforum. The researchers found that arginine hypomethylation of FUS at primary neuron axon tips decreased new protein synthesis, but when they overexpressed TNPO1, the deficits disappeared.
The work emphasizes that some of FUS’s issues, and the actions of TNPO1, have nothing to do with nuclear-cytoplasmic transport pathways. “Many people are still looking at defective cytoplasmic-to-nuclear translocation, which is clearly a problem, but additional problems occur in terminals as well,” St George-Hyslop told Alzforum.
“The underlying conclusion of this body of work is that altering the posttranslational state, and the action of chaperones, are two ways in which the cell physiologically regulates this assembly process,” said St George-Hyslop. “It’s going to need now a little bit of work to try to figure out exactly who the partners are—what are the methyltransferases, the demethylases, the kinases, the phosphatases, and the chaperones other than TNPO1, and most importantly, what regulates them,” he said. “Somewhere in that currently unclear mist of factors, we should be able to find something that can potentially be manipulated.”
Role of RNA Sequence
Turning to RNA, Alberti and Hyman were puzzled why proteins like FUS and TDP-43 aggregate in the cytosol, but remain soluble in the nucleus, where their concentrations are quite high. The answer, they found, was RNA. First author Shovamayee Maharana first estimated concentrations of FUS and other RBPs in the nucleus, then showed that, at that same concentration in vitro, 7.6 μM, FUS phase-separated. But when the researchers repeated the experiment in the presence of total cell RNA, they got a different result. As expected, low concentrations of RNA promoted liquid drop formation, but higher concentrations, as in the nucleus, suppressed it.
FUS-ion. Microinjecting HeLa cell nuclei with RNase causes immediate condensation of GFP-FUS (green) into liquid-like droplets. Arrowheads show where smaller FUS assemblies fuse into larger spherical bodies. The cell at lower right was not injected with RNase. [From Maharana et al., 2018. Courtesy of Science/AAAS.]
To test whether RNA prevents phase separation of RBPs in vivo, the researchers microinjected RNase into HeLa cell nuclei. Almost immediately, FUS and other RBPs condensed into liquid-like droplets (see movie). Injecting concentrated FUS had the same effect. RNA also retarded aberrant aggregation of FUS into fibrils, and FUS mutants with impaired RNA binding aggregated more readily and were more toxic to cells. The results suggest that high RNA in the nucleus buffers against aggregation of RBPs, the authors conclude. When RBPs move into the cytoplasm, as they do in times of stress, they become prone to aggregate, and anything that prolongs time spent in the cytosol will increase their chances of aggregation, say the authors. Based on their results, they predict that changes in RNA levels or the ability of proteins to bind RNA are frequent causes of age-related protein-misfolding diseases.
“It’s a neat setup, where each compartment has its own system for dealing with these proteins,” said Shorter. “The Kapβ2 TNPO1 in cytoplasm stops FUS from getting into trouble in that compartment, and when FUS is back in the nucleus, the high concentration of RNA prevents aggregation.”
RNA has another important function in dictating the individual identities of phase-separated membraneless organelles, says the final study. First author Erin Langdon and coworkers from the Gladfelter lab demonstrated that its three-dimensional structure dictates if RNA will get recruited into droplets in cells. Proteins play a role in determining the conformation of RNA, so are active participants in the process, they show. This mechanism is likely relevant for sorting of specific RNAs to stress granules, P bodies, and other individualized droplets. Gladfelter emphasized that it’s too early to speculate what this might have to do with neurodegenerative disease. “The field is still young—so much remains unknown about normal granule assembly, it’s impossible to say yet what might go wrong in disease,” she told Alzforum.—Pat McCaffrey
References
News Citations
Paper Citations
- Zhang ZC, Chook YM. Structural and energetic basis of ALS-causing mutations in the atypical proline-tyrosine nuclear localization signal of the Fused in Sarcoma protein (FUS). Proc Natl Acad Sci U S A. 2012 Jul 24;109(30):12017-21. Epub 2012 Jul 9 PubMed.
- Li P, Banjade S, Cheng HC, Kim S, Chen B, Guo L, Llaguno M, Hollingsworth JV, King DS, Banani SF, Russo PS, Jiang QX, Nixon BT, Rosen MK. Phase transitions in the assembly of multivalent signalling proteins. Nature. 2012 Mar 7;483(7389):336-40. PubMed.
Further Reading
Primary Papers
- Yoshizawa T, Ali R, Jiou J, Fung HY, Burke KA, Kim SJ, Lin Y, Peeples WB, Saltzberg D, Soniat M, Baumhardt JM, Oldenbourg R, Sali A, Fawzi NL, Rosen MK, Chook YM. Nuclear Import Receptor Inhibits Phase Separation of FUS through Binding to Multiple Sites. Cell. 2018 Apr 19;173(3):693-705.e22. PubMed.
- Guo L, Kim HJ, Wang H, Monaghan J, Freyermuth F, Sung JC, O'Donovan K, Fare CM, Diaz Z, Singh N, Zhang ZC, Coughlin M, Sweeny EA, DeSantis ME, Jackrel ME, Rodell CB, Burdick JA, King OD, Gitler AD, Lagier-Tourenne C, Pandey UB, Chook YM, Taylor JP, Shorter J. Nuclear-Import Receptors Reverse Aberrant Phase Transitions of RNA-Binding Proteins with Prion-like Domains. Cell. 2018 Apr 19;173(3):677-692.e20. PubMed.
- Hofweber M, Hutten S, Bourgeois B, Spreitzer E, Niedner-Boblenz A, Schifferer M, Ruepp MD, Simons M, Niessing D, Madl T, Dormann D. Phase Separation of FUS Is Suppressed by Its Nuclear Import Receptor and Arginine Methylation. Cell. 2018 Apr 19;173(3):706-719.e13. PubMed.
- Qamar S, Wang G, Randle SJ, Ruggeri FS, Varela JA, Lin JQ, Phillips EC, Miyashita A, Williams D, Ströhl F, Meadows W, Ferry R, Dardov VJ, Tartaglia GG, Farrer LA, Kaminski Schierle GS, Kaminski CF, Holt CE, Fraser PE, Schmitt-Ulms G, Klenerman D, Knowles T, Vendruscolo M, St George-Hyslop P. FUS Phase Separation Is Modulated by a Molecular Chaperone and Methylation of Arginine Cation-π Interactions. Cell. 2018 Apr 19;173(3):720-734.e15. PubMed.
- Mikhaleva S, Lemke EA. Beyond the Transport Function of Import Receptors: What's All the FUS about?. Cell. 2018 Apr 19;173(3):549-553. PubMed.
- Maharana S, Wang J, Papadopoulos DK, Richter D, Pozniakovsky A, Poser I, Bickle M, Rizk S, Guillén-Boixet J, Franzmann T, Jahnel M, Marrone L, Chang YT, Sterneckert J, Tomancak P, Hyman AA, Alberti S. RNA buffers the phase separation behavior of prion-like RNA binding proteins. Science. 2018 Apr 12; PubMed.
- Langdon EM, Qiu Y, Ghanbari Niaki A, McLaughlin GA, Weidmann C, Gerbich TM, Smith JA, Crutchley JM, Termini CM, Weeks KM, Myong S, Gladfelter AS. mRNA structure determines specificity of a polyQ-driven phase separation. Science. 2018 Apr 12; PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Massachusetts General Hospital, Harvard Medical School, German Center for Neurodegenerative Diseases (DZNE Berlin)
Six new papers in Cell and Science on droplets in very different eukaryotic systems—from mammalian cells to fungi—and involved in different cellular mechanisms. What a week!
These new works all point at the complexity of LLPS in living systems and show us the mechanisms beyond protein concentrations and modifications; they present us examples of how heteromeric interactions of biomolecules in liquid droplets can determine droplet function and how this is used by cells as solutions to regulate different essential processes in the cell, such as the nuclear-cytoplasmic transport, spatial organization and restriction of droplet formation, and RNA selectivity and droplet specification.
The two papers published in Science focus on the so far underappreciated (or at least under-addressed) role of RNA in the process of mammalian stress granule formation (Maharana et al., from the Alberti Lab) and in the dynamics and spatial separation of droplets involved in distinct cell functions in fungi (Langdon et al., from the Gladfelter Lab). Although addressing two absolutely different processes, the two papers do have a lot in common: They show that not only proteins and protein modifications play a role in RNA-binding protein biology and pathology, but introduce also the pivotal role of RNA kind, structure, and sequence in these processes. That the specifics of RNA play a big role in RNA-binding protein LLPS is not surprising, however, we now learn that not necessarily the proteins and their structure “select” the RNA, but that RNA structure and hybridization has important scaffolding activity for protein droplets and “select” the proteins—we may as well call them “RNA-protein droplets.”
One very important message delivered by these two new RNA-protein LLPS papers is the notion that the complex interplay between proteins and RNA in cellular LLPS exceeds our often protein-central view. They connect the protein world with the RNA world beyond translation and transcription, and thereby offer a fresh view on the role of different RNA species beyond encoding (mRNA) and facilitating (tRNA) protein production. With this, RNA homeostasis gets a fresh look in biology and health/disease, and a novel role of the vast amount of noncoding RNAs evolves. I am excited to see how we can embed these concepts in the field of neurodegenerative diseases such as ALS and Alzheimer’s disease, in which LLPS has been suggested to play a role for disease initiation and progression.
Thank you for these inspiring works!
Neurobiology, KU Leuven & VIB
Overall, the different Cell papers nicely document how nuclear import proteins function as chaperones: not only to transport disease-related RNA-binding proteins through the nuclear pore, but also to suppress unwanted liquid-liquid phase separations of these proteins into stress granules (for a recent review on liquid-liquid phase separations, see Boeynaems et al., 2018). Both defects in nucleocytoplasmic transport, as well as disturbances in liquid-liquid phase separations, have recently been linked by many research groups to amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD). Defects in both processes not only can explain how certain important disease-related proteins become mislocalized into the cytoplasm, but also provide a framework to understand the generation of aggregates. Proteins present in the droplets can undergo an irreversible liquid-to-solid phase transition, which ultimately results in the formation of solid, fibrous aggregates observed in postmortem tissue of patients.
The concept that nuclear import receptors function specifically as chaperones for wild-type and disease-linked RNA-binding proteins with a nuclear localization signal is convincingly shown by Guo et al. Karyopherin-β2/Transportin-1 interacts with the PY-nuclear localization signal (NLS) to inhibit and/or reverse FUS, TAF15, EWSR1, hnRNPA1, and hnRNPA2 fibrillization. Most remarkably, Karyopherin-β2/Transportin-1 can also restore the nuclear localization of these proteins in yeast and patient fibroblasts. Interestingly, Importin-α in combination with Karyopherin-β1 performs a similar function in relation to TDP-43, and these proteins can also prevent and reverse TDP-43 fibrillization.
What makes these studies extremely interesting is the fact that a better insight is also provided into the way the nuclear import proteins are interacting with their cargos. Again, the focus is mainly on FUS and Karyopherin-β2/Transportin-1. Apart from the tight binding to the C-terminal nuclear localization signal of FUS, Yoshizawa et al. show that Karyopherin-β2/Transportin-1 disturbs the liquid-liquid phase separation by weak interactions of this importin with sequences distributed throughout FUS. Not unexpectedly, the N-terminal low-complexity domain (also called the prion-like domain) of FUS is involved in these weak interactions. Interestingly, also other domains like the RGG regions, the RRM and the zinc finger (ZnF) domain play important roles in these interactions. The effect of Karyopherin-β2/Transportin-1 on the liquid-liquid phase separation of FUS seems to be due to a competition with the FUS-FUS interactions.
Further important mechanistic insights are provided by Hofweber et al. and Qamar et al. They show that not only Karyopherin-β2/Transportin-1 but also arginine methylation of FUS have a function beyond nuclear import. This could be extremely important in the context of FTD as mislocalization and aggregation of FUS is observed in a subpopulation of FTD patients in the absence of FUS mutations. Both Karyopherin-β2/Transportin-1 and arginine methylation suppress liquid-liquid phase separation and stress granule association of FUS. These papers also add further convincing evidence to the concept that the N-terminal prion-like domain of FUS is not only important for liquid-liquid phase separation. As is clearly shown, the RGG domains and especially the arginines in these motifs also play crucial roles in liquid-liquid phase separation of FUS. These findings are in line with previous reports that the RGG-ZnF-RGG domains of FUS can form fibrous assemblies in vitro (Schwartz et al., 2013) and with our recent observation that synthetic FUS-RGG peptide forms droplets in the presence of a molecular crowder or polyU RNA (Boeynaems et al., 2017).
Overall, the observations reported here nicely complement our recent observations that the arginine-rich dipeptide repeat proteins (polyPR and polyGR) which are translated from the hexanucleotide repeats in C9ORF72 can also undergo liquid-liquid phase separations (Boeynaems et al., 2017). These dipeptide repeat proteins can bind to many proteins with low-complexity domains and can influence the liquid-liquid phase separation of arginine-rich proteins with low complexity domains, including FUS.
Now that the role of nuclear import receptors in the liquid-liquid phase separation of different disease-related RNA-binding proteins is firmly established, the next step will be to obtain more evidence that these mechanisms are also at play in more complex disease-related in vivo models. However, the biggest challenge will be to interfere therapeutically with the liquid-liquid phase separation process. As well as nucleocytoplasmic transport, it is a fundamental biological process. A controlled and cell-specific interference with such general and crucial processes will be extremely challenging. One doesn’t want to make things worse!
References:
Boeynaems S, Alberti S, Fawzi NL, Mittag T, Polymenidou M, Rousseau F, Schymkowitz J, Shorter J, Wolozin B, Van Den Bosch L, Tompa P, Fuxreiter M. Protein Phase Separation: A New Phase in Cell Biology. Trends Cell Biol. 2018 Mar 27; PubMed.
Boeynaems S, Bogaert E, Kovacs D, Konijnenberg A, Timmerman E, Volkov A, Guharoy M, De Decker M, Jaspers T, Ryan VH, Janke AM, Baatsen P, Vercruysse T, Kolaitis RM, Daelemans D, Taylor JP, Kedersha N, Anderson P, Impens F, Sobott F, Schymkowitz J, Rousseau F, Fawzi NL, Robberecht W, Van Damme P, Tompa P, Van Den Bosch L. Phase Separation of C9orf72 Dipeptide Repeats Perturbs Stress Granule Dynamics. Mol Cell. 2017 Mar 16;65(6):1044-1055.e5. PubMed.
Schwartz JC, Wang X, Podell ER, Cech TR. RNA seeds higher-order assembly of FUS protein. Cell Rep. 2013 Nov 27;5(4):918-25. Epub 2013 Nov 21 PubMed.
Make a Comment
To make a comment you must login or register.