The Chaperone FKBP12 Shields Tau from Aggregation
Quick Links
The 12 kilodalton FK506 binding protein (FKBP12) interacts with tau to attenuate aggregation. But how this small protein filled such big shoes was unclear—until now. In the February 1 Science Advances, researchers led by Benjamin Wolozin at Boston University and Markus Zweckstetter of the German Center for Neurodegenerative Diseases in Göttingen reported that FKBP12 binds to two specific motifs within tau that are aggregation-prone when phosphorylated. Overexpressing FKBP12 tempered insoluble tau in cultured neurons seeded with tau oligomers and reduced neurodegeneration in three-dimensional cultures of human neurons and astrocytes.
- FKBP12 binds to two aggregate-prone segments of tau.
- Overexpressing the chaperone protein halves tau aggregation in mouse neurons.
- FKBP12 quells tau oligomerization and neurodegeneration in three-dimensional human neuron-astrocyte cultures.
“This is a very impressive study suggesting that FKBP12 is neuroprotective against tau-related pathology,” Kun Ping Lu, University of Western Ontario, Canada, told Alzforum.
Like other FKBPs, FKBP12 has dual functions: as a prolyl isomerase that flips the cyclic amino acid proline between its cis and trans forms to alter a protein’s structure, and as a chaperone to help fold intrinsically disordered proteins, such as tau (Feb 2010 news). Notably, FKBP12 is depleted in Alzheimer’s disease brain tissue yet enriched in neurofibrillary tangles (Sugata et al., 2009). The chaperone also prevented aggregation of a tau peptide spanning residues 306 to 336 in the protein’s third repeat segment, and its isomerase activity was essential (Ikura and Ito, 2013). Wolozin and Zweckstetter wondered how FKBP12 kept tau from aggregating.
To address this, Zweckstetter turned to the electron microscope. He saw shorter tau fibrils had formed in vitro when FKBP12 was present, suggesting that the protein slows elongation, probably by binding to tau.
Where on tau might FKBP12 bind? Using hydrogen, carbon, and nitrogen NMR, co-first author Pijush Chakraborty found that tau remained mostly disordered in the presence of the chaperone protein. However, a spike in signal intensity of two short segments, residues 307 to 311 and 391 to 395, indicated a more stable structure, suggesting FKBP12 might bind there.
These motifs, QIVYK and EIVYK, have almost identical sequences and similar three-dimensional structures, as determined by NMR spectra or by high-resolution models of the tau-FKBP12 complex generated by the protein-folding software AlphaFold (see image below). The authors were struck by how specific FKBP12’s binding was to the (Q/E)IVYK motif. “This specificity isn’t seen with other FKBPs,” said co-author Laura Blair of the University of Southern Florida, Tampa. Intriguingly, molecular modeling showed QIVYK and EIVYK snuggled into the catalytic pocket of FKBP12 despite neither having a proline residue.
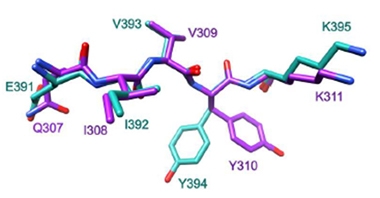
FKBP12 Motifs. Two tau sequences, QIVYK (purple) and EIVYK (cyan), assume almost identical structures upon binding FKBP12. [Courtesy of Jiang et al., Science Advances, 2023.]
Could the tyrosine residues within these motifs be critical for FKBP12 modulation of tau aggregation, since that amino acid is often phosphorylated and hyperphosphorylation of tau drives its aggregation? Indeed, after phosphorylating Y310 and Y394 in human tau monomers, neither QIVYK nor EIVYK NMR signals changed after adding FKBP12, indicating that phosphorylation blocked binding.
Zweckstetter wondered if FKBP12 prevents tau aggregation by shielding those tyrosine residues from phosphorylation, though the authors did not test this. Wolozin was intrigued that the chaperone acts on tau monomers to prevent their aggregation, since, as Blair noted, FKBP51 and FKBP52 do the opposite, promoting tau oligomerization and fibrillization, respectively (Sep 2013 news; Criado-Marrero et al., 2021). “Even though FKBPs are closely related, they all have their unique actions,” she said.
Could FKBP12 temper tau aggregation or even neurotoxicity? Indeed, when co-first author Lulu Jiang in the Wolozin lab overexpressed the chaperone in cultured mouse neurons expressing human tau, it reduced phospho-tau and cleaved caspase 3, a neuronal apoptosis marker, induced by tau oligomers. Knocking out FKBP12 did the opposite.
Jiang saw the same protection when she overexpressed FKBP12 in three-dimensional human neuron-astrocyte cultures that produce phosphorylated tau aggregates, reactive astrocytes, and neurodegeneration after seeding with tau oligomers (Rickner et al., 2022). She saw less phosphorylated, misfolded tau and fewer dying neurons (see image below).

More FKBP12, Less Pathology. When 3D human neuron-astrocyte cultures (top) were seeded with oligomers of human tau, they produced their own toxic tau oligomers (red), and neurons (purple) degenerated as judged by Fluoro-Jade B staining (green). Overexpressing FKBP12 (bottom) reduced oligomer formation and protected neurons. [Courtesy of Jiang et al., Science Advances, 2023.]
All told, the authors concluded that FKBP12 plays a critical role in regulation of tau pathology. They noted that this does not rule out other neuroprotective roles, since FKBP12 binds many substrates.—Chelsea Weidman Burke
References
News Citations
- Shall We Dance? Isomerases Twist With Tau, Synuclein, Ca2+ Channels
- Chaperone “Saves” Tau, Turning it into Toxic Oligomers
Mutation Interactive Images Citations
Paper Citations
- Sugata H, Matsuo K, Nakagawa T, Takahashi M, Mukai H, Ono Y, Maeda K, Akiyama H, Kawamata T. A peptidyl-prolyl isomerase, FKBP12, accumulates in Alzheimer neurofibrillary tangles. Neurosci Lett. 2009 Aug 7;459(2):96-9. PubMed.
- Ikura T, Ito N. Peptidyl-prolyl isomerase activity of FK506 binding protein 12 prevents tau peptide from aggregating. Protein Eng Des Sel. 2013 Sep;26(9):539-46. PubMed.
- Criado-Marrero M, Gebru NT, Gould LA, Blazier DM, Vidal-Aguiar Y, Smith TM, Abdelmaboud SS, Shelton LB, Wang X, Dahrendorff J, Beaulieu-Abdelahad D, Dickey CA, Blair LJ. FKBP52 overexpression accelerates hippocampal-dependent memory impairments in a tau transgenic mouse model. NPJ Aging Mech Dis. 2021 May 3;7(1):9. PubMed.
- Rickner HD, Jiang L, Hong R, O'Neill NK, Mojica CA, Snyder BJ, Zhang L, Shaw D, Medalla M, Wolozin B, Cheng CS. Single cell transcriptomic profiling of a neuron-astrocyte assembloid tauopathy model. Nat Commun. 2022 Oct 21;13(1):6275. PubMed.
External Citations
Further Reading
Primary Papers
- Jiang L, Chakraborty P, Zhang L, Wong M, Hill SE, Webber CJ, Libera J, Blair LJ, Wolozin B, Zweckstetter M. Chaperoning of specific tau structure by immunophilin FKBP12 regulates the neuronal resilience to extracellular stress. Sci Adv. 2023 Feb 3;9(5):eadd9789. Epub 2023 Feb 1 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University of Texas, Southwestern Medical Center
This is a solid paper in terms of biochemistry/biophysics. The authors document an interaction between tau and FKBP in vitro, and specify the interaction mode. It is possible that the FKBP is binding and regulating tau in cells, but the co-localization experiments are not conclusive. I would have liked to see, e.g., an immunoprecipitation showing a direct interaction in cells. One thing the authors could have done would be to modify amino acids in tau that change binding to FKBP and then test if that alters the rescue effects of FKBP. FKBP binds a lot of substrates, as they note, thus many of the protective effects they assert could easily be indirect, and not related to binding to tau.
The University of Minnesota
It is well known in the field that by inhibiting tau-mediated pathologies and synaptic dysfunction, FK506 is protective in neurodegenerative diseases, including frontotemporal dementia (Yoshiyama et al., 2007), Alzheimer’s disease (Miller et al., 2014) and Parkinson’s disease (Teravskis et al., 2018). FKBP12 is one of the best-characterized receptors of FK506, which was initially developed as an immunosuppressive drug but was found to be a strong inhibitor of long-term depression (LTD), a form of synaptic plasticity, as well as a rescuer of pathological forms of LTD. How, was unclear. The reported direct interaction between FKBP12 and tau will help solve this decade-long puzzle in the field.
The authors reported that FKBP12 binds to a unique structural motif of tau and that its overexpression protects against tau pathologies and prevents neurodegeneration in a tauopathy model. This significantly advances the field by providing a novel mechanism by which FK506 inhibits tau pathologies and tau-mediated pathophysiology. Moreover, tau mislocalization from axons to postsynaptic structures including the somatodendritic domain is believed to be a common mechanism underlying the pathobiology of multiple neurodegenerative disease. The upstream signaling cascade that causes this tau mislocalization remains elusive. The reported oligomeric tau-induced co-mislocalization of FKBP12 and tau to the somatodendritic domain may also shed new light upon this unresolved puzzle. Nevertheless, although highly novel, this interaction between tau and FKBP12 provides limited information for how tau, FKBP12, and calcineurin mediate tau pathophysiology in neurodegenerative diseases. Further studies are needed to determine whether FKBP12 acts upstream to or downstream from tau and calcineurin.
References:
Yoshiyama Y, Higuchi M, Zhang B, Huang SM, Iwata N, Saido TC, Maeda J, Suhara T, Trojanowski JQ, Lee VM. Synapse loss and microglial activation precede tangles in a P301S tauopathy mouse model. Neuron. 2007 Feb 1;53(3):337-51. PubMed.
Miller EC, Teravskis PJ, Dummer BW, Zhao X, Huganir RL, Liao D. Tau phosphorylation and tau mislocalization mediate soluble Aβ oligomer-induced AMPA glutamate receptor signaling deficits. Eur J Neurosci. 2014 Apr;39(7):1214-24. PubMed.
Teravskis PJ, Covelo A, Miller EC, Singh B, Martell-Martínez HA, Benneyworth MA, Gallardo C, Oxnard BR, Araque A, Lee MK, Liao D. A53T Mutant Alpha-Synuclein Induces Tau-Dependent Postsynaptic Impairment Independently of Neurodegenerative Changes. J Neurosci. 2018 Nov 7;38(45):9754-9767. Epub 2018 Sep 24 PubMed.
Make a Comment
To make a comment you must login or register.