Brain Anatomy Revealed With CLARITY
Quick Links
Adapted from a story that originally appeared on the Schizophrenia Research Forum.
10 April 2013. A method for examining the detailed structures of the intact brain with light microscopy debuts in a paper coming out April 11 in Nature. Called CLARITY, and developed by Karl Deisseroth and colleagues at Stanford University, Palo Alto, California, the technique washes away the fatty cell membranes of neurons while leaving in place the rest of the cell’s parts, including proteins and nucleic acids. This rendered intact mouse brains transparent and permeable, which allowed an unobstructed look at neurons and their processes with no need for sectioning or reconstruction. The technique also worked with human postmortem samples preserved for years in fixative, and thus could help reveal an integrated view of brain organization in disorders such as Alzheimer's disease and schizophrenia. The advance made the homepage of The New York Times.
Until now, the brain has given up the details of its organization one thin slice at a time. Because light from microscopes cannot penetrate very far into the brain before getting refracted by the lipid bilayers of cell membranes, researchers cut the brain into slices tens of microns thin. These slices also expose components of the cell so that they can be labeled with markers, such as fluorescent antibodies. After generating images of each slice, researchers then reconstruct portions of the intact brain by realigning the images.
The new technique circumvents this painstaking process. Recognizing that lipid bilayers both scatter light and interfere with access to cell components, the researchers got rid of the membranes while preserving structure with a stand-in scaffolding onto which cellular constituents were chemically tethered. This left behind a porous, yet fully assembled system of cell parts, amenable to imaging and labeling of different cellular molecules. Though other intact-imaging techniques have extended the depth of light microscopy (e.g., Hama et al., 2011), they do not offer a preparation permeable enough for labeling.
Call it CLARITY
First author Kwanghun Chung and colleagues began by perfusing mouse brains with a mixture of formaldehyde and hydrogel monomers. Formaldehyde, a traditional fixation chemical, links molecules together to preserve tissue structure. In this preparation, formaldehyde molecules linked cell parts such as proteins or nucleic acids to the hydrogel monomers, which then polymerized to form a lattice-like mesh. This provided a scaffold upon which cell parts were firmly linked when the lipid bilayers were removed. A specially designed chamber applied an electric field across the brain to expedite the lipid exit, leaving behind a transparent brain. In all, the process took about eight days.
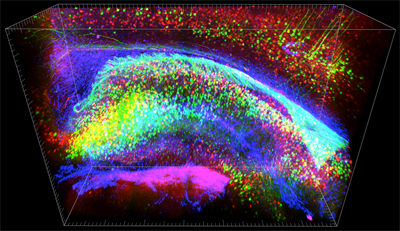
The researchers examined the brain of an adult mouse expressing a fluorescent protein in a subset of projection neurons. In a 3.4 mm cube of tissue, the microscope revealed the axonal pathways traversing the brain, a layer of cortical neurons with their dendrites stretching above them, and at higher magnification, the spines along these dendrites. There did not seem to be any loss of resolution deeper into the sample, so structures of the brainstem, thalamus, hippocampus, and cortex were equally visible.
Multiple Rounds of Molecular Phenotyping
Because this transparent brain is also permeable, the researchers labeled different cell parts, a process they called “molecular phenotyping.” CLARITY degraded significantly less protein than conventional methods of clearing tissue. Indeed, immunohistochemical labeling identified tyrosine hydroxylase (TH), a key enzyme for making catecholamines such as dopamine, throughout the cleared brain, including in deep regions such as the ventral tegmental area and dorsal raphe nucleus. To reach into the depths of an intact brain, however, required a six-week-long incubation and wash process—significantly longer than immunohistochemistry with sectioned tissue.
Three-dimensional view of intact hippocampal tissue block showing enhanced yellow fluorescent protein-expressing neurons (green), parvalbumin-positive neurons (red), and glial fibrillary acidic protein GFAP (blue). Image courtesy of Kwanghun Chung and Karl Deisseroth
Labeling for an assortment of other molecules worked, too, and this was expedited by cutting the intact brain into a 500-micron block. Three days of immunolabeling for synapsin, a presynaptic marker, and PSD-95, a postsynaptic marker, revealed individual excitatory synaptic contacts. The researchers also labeled small non-protein molecules such as γ-aminobutyric acid and messenger RNA. The researchers also found that the CLARITY-treated brain could withstand multiple rounds of labeling. For example, one round of TH-labeling, followed by washing it away, followed by another round of TH-labeling, produced identical patterns of TH signal.
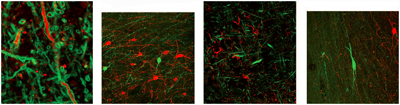
Immunohistological visualization and identification of neurons and projections in postmortem tissue. Image courtesy of Kwanghun Chung and Karl Deisseroth
Take It to the Brain Banks
The researchers also found that CLARITY worked with precious human brain samples already in storage. They cut a 500-micron-thick slice from a postmortem frontal lobe sample from a person with autism that had been stored for over six years in formalin, and treated it with the CLARITY procedure. After staining with antibodies, the researchers could visualize and trace individual axons. By staining for parvalbumin, a protein in certain kinds of inhibitory neurons that are suspected of contributing to imbalances in excitatory and inhibitory signaling in autism and schizophrenia (see SRF related news story), they visualize these neurons embedded within the cortex, along with the full extent of their processes. Curiously, some of these processes formed ladder-like “dendritic bridges,” either by reconnecting with themselves or with processes from other neurons. A postmortem sample from a control brain did not reveal as many of these ladders.—Michele Solis
References
Paper Citations
- Hama H, Kurokawa H, Kawano H, Ando R, Shimogori T, Noda H, Fukami K, Sakaue-Sawano A, Miyawaki A. Scale: a chemical approach for fluorescence imaging and reconstruction of transparent mouse brain. Nat Neurosci. 2011 Nov;14(11):1481-8. PubMed.
Other Citations
External Citations
Further Reading
Primary Papers
- Chung K, Wallace J, Kim SY, Kalyanasundaram S, Andalman AS, Davidson TJ, Mirzabekov JJ, Zalocusky KA, Mattis J, Denisin AK, Pak S, Bernstein H, Ramakrishnan C, Grosenick L, Gradinaru V, Deisseroth K. Structural and molecular interrogation of intact biological systems. Nature. 2013 Apr 10; PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.