Aβ Binds Glutamate Receptors. Only in Guys, Though.
Quick Links
Could a person’s sex dictate how his or her brain reacts to amyloid? Stephen Ferguson, University of Ottawa Brain and Mind Institute, Canada, thinks it may. In a paper published December 15 in Science Signaling, his group reported that Aβ bound to the mGluR5 metabotropic glutamate receptor in postmortem brain tissue from men but not women. The scientists found the same in mice. Why might that be? It turns out that mGluR5 and the cellular prion protein, which form a trimeric complex with Aβ, do not bind each other in the female brain. The reason for this binding deficiency remains elusive. If confirmed, it would highlight the importance of sex-specific biology for understanding and treating Alzheimer’s disease.
- Aβ binds metabotropic glutamate receptors in males but not females.
- Estrogen does not explain the difference, but cellular prion protein may.
- PrP-Aβ-mGluR complexes form only in male mice.
“I was fascinated—I would never expect these fundamental protein mechanisms to be different in male and female brains,” said Helmut Kessels, University of Amsterdam, Netherlands.
Glutamate, an excitatory neurotransmitter, plays a central role in learning and memory. In neurons, glutamate activates ionotropic NMDA and AMPA receptors and metabotropic receptors such as mGluR5. The latter also binds Aβ oligomers and the cellular prion protein (PrPc). Some scientists believe this troika is neurotoxic (Feb 2009 conference news; Jun 2010 news). Evidence suggests the tripartite complex activates a toxic cascade that accelerates Aβ aggregation, compromises autophagy, and shrivels synapses (Sep 2013 news; Oct 2015 news).

Men Only? Aβ oligomers bind to cellular prion protein and mGluR5, triggering downstream problems—but only in one sex. [Courtesy of Abd-Elrahman et al., Science Signaling, 2020.]
Previously, Ferguson’s group inhibited mGluR5 and found this reduced Aβ pathology and protected against cognitive decline in mouse models of amyloidosis (May 2016 news; Abd-Elrahman et al., 2018). Because the scientists had only studied male mice, they wondered if females with amyloid were equally protected.
Co-first authors Khaled Abd-Elrahman and Awatif Albaker initially turned to wild-type mice to see if Aβ bound to mGluR5 in male and female mouse brains equally well. As expected, synthetic Aβ oligomers displaced, in a dose-dependent manner, the radiolabeled mGluR5 antagonist MPEP from plasma cell membranes prepared from 3-month-old male cortical or hippocampal tissue. However, the oligomers did not budge the antagonist from female cell membranes, indicating the peptide does not bind their mGluR5. Likewise, Aβ displaced the antagonist from cell membranes prepared from postmortem brain tissue from men, but not from women. “That Aβ42 did not bind at all to either mouse or human female mGluR5 receptors was completely shocking,” Ferguson said.
One obvious difference between men and women are sex hormones. To find out whether estrogen was involved in this odd “gender discrimination,” Abd-Elrahman injected 3-month-old, wild-type female mice each day for two weeks with fulvestrant. This drug suppresses estrogen receptor expression and is used to treat certain types of breast cancer. Then, the researchers repeated the previous binding experiments. The estrogen antagonist did not change Aβ-mGluR5 binding (see image below).

Don’t Blame Estrogen. In a competition experiment, the mGluR5 antagonist MPEP (open circles) displaces radiolabeled MPEP from female cortical cell membranes, whether or not the animals had been treated with the estrogen antagonist fulvestrant (pink). Aβ (closed circles) failed to displace MPEP, even after fulvestrant treatment. [Courtesy of Abd-Elrahman et al., Science Signaling, 2020.]
Looking for the next logical explanation, the researchers turned to the Aβ co-receptor, PrPc. Although levels were similar in 3-month-old, wild-type male and female brains, prion protein only co-immunoprecipitated with mGluR5 from male tissue. The findings indicate that the mGluR5-PrPc complex did not form in female mouse brain tissue.
This puzzled Kessels. “Perhaps another protein is needed for the PrPc-mGluR5 interaction [in females],” he said. Michael Rowan, Trinity College Dublin, favored a different explanation. “I think the difference may be caused by a sexual dimorphism in the ability of [endogenous] Aβ-bound PrP to couple to mGluR5 rather than a difference in mGluR5 affinity for Aβ,” he wrote.
Rowan noted that Aβ and PrPc form complexes in the brains of men and women who have AD but not in healthy controls (Dohler et al., 2014). “Taken together, these findings raise the intriguing possibility that female pathology and cognitive impairment in Alzheimer’s disease is driven, at least partly, by pathogenic partners for Aβ-PrP complexes other than mGluR5,” he wrote to Alzforum (see full comment below). But Rowan also cautioned that none of the four female donors had any evidence of cognitive impairment or brain pathology, whereas two of the four male donors had dementia with Lewy bodies.
Aβ oligomers binding to mGluR5 slows down autophagy, and this can be reversed by blocking the mGluR5 (Abd-Elrahman et al., 2018). To see if this downstream effect is also sex-specific, the authors tested primary cortical neurons derived from wild-type mouse embryos. After the researchers established the primary neurons, they added Aβ synthetic oligomers. Autophagy slowed in male neurons only.
APP/PS1 mice told the same story. Six-month-old males, but not females, accumulated more p62, a sign of dysfunctional autophagy, than did wild-type animals. To see if autophagy could be improved, the researchers treated the male mice with the mGluR5 negative allosteric modulator CTEP. After 12 weeks, p62 levels had dropped to normal.
Earlier this year, the same scientists reported that treating 6-month-old male APP/PS1 mice with CTEP for 24 weeks reduced plaque load and improved autophagy (Abd-Elrahman et al., 2020). They had also seen fewer plaques and better cognitive function in 9-month-old APP/PS1 and 3xTg male mice treated with CTEP (May 2016 news). In the current study, they report about 40 percent reduction of plaques in the hippocampus and cortex, but again only in males. They also found that the male APP/PS1 mice, but not females, better recognized novel objects and remembered the location of a hidden platform in a water maze when treated with the modulator.
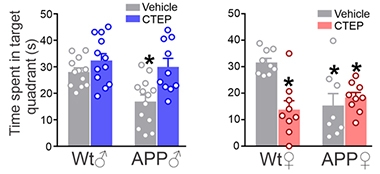
Double Whammy for Females. After 12 weeks on the mGluR5 agonist CTEP, male APP/PS1 mice better remembered the location of a hidden platform in a water maze, spending more time searching the correct quadrant than did untreated mice (left). In females (right), CTEP treatment failed to improve the performance of the APP/PS1 mice and worsened that of wild-type. [Abd-Elrahman et al., Science Signaling, 2020.]
These findings come with some curious anomalies. In their paper from earlier this year, the authors described how the benefit of CTEP waned after 36 weeks. “At a certain age, mGluR5 is not contributing to the disease anymore. The aged male mice look just like female mice in terms of cell signaling, with no improved autophagy,” Ferguson said. This hints that mGluR5 responses may only be important early in disease.
In another twist, CTEP weakened memory in wild-type females, which took longer to escape the water maze than untreated controls. CTEP had no effect on wild-type males (see image above). Kessels found this curious. “It is surprising that memory was only impaired in females. I would expect this drug to impair memory in both males and females because mGluR5 enhances synaptic plasticity, which is needed for learning,” he said.
Still, the findings point to the importance of studying sex-specific differences in AD and in AD drug discovery. “Maybe drugs have failed in neurodegenerative diseases because we have not looked at the right populations; some may be more effective in men than women or vice versa,” Ferguson said. “Why the Aβ-PrPc-mGluR5 complex only forms in males remains a mystery,” he added.—Chelsea Weidman Burke
References
News Citations
- Keystone: Partners in Crime—Do Aβ and Prion Protein Pummel Plasticity?
- Aβ Oligomers: A Fatal Attraction for Glutamate Receptors?
- Glutamate Receptor Links Aβ-Prion Complex with Fyn, Synaptic Damage
- In APP Knock-Ins, Calcium Chaos Dismantles Mushroom Spines
- Shielding Synaptic Glutamate Receptor from Aβ Preserves Memory in Mice
Research Models Citations
Paper Citations
- Abd-Elrahman KS, Hamilton A, Vasefi M, Ferguson SS. Autophagy is increased following either pharmacological or genetic silencing of mGluR5 signaling in Alzheimer's disease mouse models. Mol Brain. 2018 Apr 10;11(1):19. PubMed.
- Dohler F, Sepulveda-Falla D, Krasemann S, Altmeppen H, Schlüter H, Hildebrand D, Zerr I, Matschke J, Glatzel M. High molecular mass assemblies of amyloid-β oligomers bind prion protein in patients with Alzheimer's disease. Brain. 2014 Mar;137(Pt 3):873-86. Epub 2014 Feb 10 PubMed.
- Abd-Elrahman KS, Hamilton A, Albaker A, Ferguson SS. mGluR5 Contribution to Neuropathology in Alzheimer Mice Is Disease Stage-Dependent. ACS Pharmacol Transl Sci. 2020 Apr 10;3(2):334-344. Epub 2020 Mar 12 PubMed.
Further Reading
Primary Papers
- Abd-Elrahman KS, Albaker A, de Souza JM, Ribeiro FM, Schlossmacher MG, Tiberi M, Hamilton A, Ferguson SS. Aβ oligomers induce pathophysiological mGluR5 signaling in Alzheimer's disease model mice in a sex-selective manner. Sci Signal. 2020 Dec 15;13(662) PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
Trinity College
The finding by Abd-Elrahman and co-authors that an mGluR5 negative allosteric modulator (NAM) alleviated cognitive and autophagy disruption in male, but not female, transgenic mice modelling Alzheimer’s disease amyloidosis raises important questions. In particular, can we conclude, as the authors do, that agents targeting mGluR5 may not be relevant to treating women with Alzheimer’s disease?
To underpin their findings, they report that synthetic Aβ oligomers displaced NAM binding to mGluR5 in healthy mouse brain in a sex-dependent manner. Remarkably, they found the same sex difference in Aβ-mediated mGluR5 ligand displacement in human postmortem cerebral cortex tissue. It should be noted that the four women tissue donors had no evidence of cognitive or brain pathology, whereas three of the four male donors had brain disorders, including two cases of dementia with Lewy bodies. Although the mechanism of this differential effect of Aβ is uncertain, the apparent concordance between the human and rodent data seems to provide some validation of animal models that study the deleterious actions of exogenously applied Aβ oligomers in both male and female healthy rodent brain. The similar sexual divergence in the APP/PS1 mice hints that, at least in this model, endogenously generated Aβ mediates some key sex-dependent pathological changes in early Alzheimer’s disease amyloidosis.
Consistent with a large body of evidence that certain Aβ oligomers bind potently to specific sequences in cellular prion protein (PrP) (Purro et al., 2018; Corbett et al., 2020) and thereby interfere with mGluR5 localization and function (Brody and Strittmatter, 2018), the NAM-displacing action of Aβ in male brain was absent in preparations lacking PrP. It seems likely, therefore, that the sex differences observed by Abd-Elrahman et al. may be caused by dimorphism in the ability of Aβ-bound PrP to aberrantly couple to mGluR5s, rather than a difference in the affinity of mGluR5 itself for Aβ. Human brain Aβ forms a complex with PrP equally in both men and women with Alzheimer’s disease, but Aβ-PrP complexes appear to be absent in the brains of healthy controls of either sex (Dohler et al., 2014). Taken together, these findings raise the intriguing possibility that female pathology and cognitive impairment in Alzheimer’s disease is driven at least partly by other, non-mGluR5, pathogenic partner(s), of Aβ-PrP complexes.
Clearly Aβ has a plethora of binding sites other than PrP, many of which have the potential to mediate its deleterious actions. Both these and Aβ production/clearance may at least partly be regulated by mGluR5 (e.g. Lee et al., 1995). Whether or not these putative mGluR5-mediated mechanisms are sex-dependent still needs to be investigated. Intriguingly, brief NAM treatment of healthy zebrafish only increased motor activity and brain mGluR5 number in males (Albasanz et al., 2016). If confirmed in humans (Smart et al., 2019), this finding indicates that unwanted side effects of NAMs, such as disruption of synaptic plasticity underlying cognitive function in the healthy brain (e.g. Collingridge et al., 2010; Qi et al., 2013), may also be sexually divergent. Unlike targeting mGluR5 with NAMs, using selective positive or silent allosteric modulators may exert fewer unwanted and potentially greater therapeutic effects, including PrP-mGuR5-independent. As the song goes, There’s Life in the Old (mGluR5) Girl Yet.
References:
Albasanz JL, Santana S, Guzman-Sanchez F, León D, Burgos JS, Martín M. 2-Methyl-6-(phenylethynyl)pyridine Hydrochloride Modulates Metabotropic Glutamate 5 Receptors Endogenously Expressed in Zebrafish Brain. ACS Chem Neurosci. 2016 Dec 21;7(12):1690-1697. Epub 2016 Sep 29 PubMed.
Brody AH, Strittmatter SM. Synaptotoxic Signaling by Amyloid Beta Oligomers in Alzheimer's Disease Through Prion Protein and mGluR5. Adv Pharmacol. 2018;82:293-323. Epub 2017 Oct 25 PubMed.
Collingridge GL, Peineau S, Howland JG, Wang YT. Long-term depression in the CNS. Nat Rev Neurosci. 2010 Jul;11(7):459-73. PubMed.
Corbett GT, Wang Z, Hong W, Colom-Cadena M, Rose J, Liao M, Asfaw A, Hall TC, Ding L, DeSousa A, Frosch MP, Collinge J, Harris DA, Perkinton MS, Spires-Jones TL, Young-Pearse TL, Billinton A, Walsh DM. PrP is a central player in toxicity mediated by soluble aggregates of neurodegeneration-causing proteins. Acta Neuropathol. 2020 Mar;139(3):503-526. Epub 2019 Dec 18 PubMed.
Dohler F, Sepulveda-Falla D, Krasemann S, Altmeppen H, Schlüter H, Hildebrand D, Zerr I, Matschke J, Glatzel M. High molecular mass assemblies of amyloid-β oligomers bind prion protein in patients with Alzheimer's disease. Brain. 2014 Mar;137(Pt 3):873-86. Epub 2014 Feb 10 PubMed.
Lee RK, Wurtman RJ, Cox AJ, Nitsch RM. Amyloid precursor protein processing is stimulated by metabotropic glutamate receptors. Proc Natl Acad Sci U S A. 1995 Aug 15;92(17):8083-7. PubMed.
Purro SA, Nicoll AJ, Collinge J. Prion Protein as a Toxic Acceptor of Amyloid-β Oligomers. Biol Psychiatry. 2018 Feb 15;83(4):358-368. Epub 2017 Nov 21 PubMed.
Qi Y, Hu NW, Rowan MJ. Switching off LTP: mGlu and NMDA receptor-dependent novelty exploration-induced depotentiation in the rat hippocampus. Cereb Cortex. 2013 Apr;23(4):932-9. Epub 2012 Apr 5 PubMed.
Smart K, Cox SM, Scala SG, Tippler M, Jaworska N, Boivin M, Séguin JR, Benkelfat C, Leyton M. Sex differences in [11C]ABP688 binding: a positron emission tomography study of mGlu5 receptors. Eur J Nucl Med Mol Imaging. 2019 May;46(5):1179-1183. Epub 2019 Jan 9 PubMed.
University of Arkansas for Medical Sciences
It is worth noting that the responses of mice to scrapie forms of PrP are reportedly sex-dependent (Loeuillet et al., 2010). In that case, greater virulence (a shorter incubation period) was observed in females, but it is easy to imagine that the structural requirements for scrapie propagation are distinct from those that permit this Aβ-mGluR5 interaction. The sex-dependency of scrapie latency appears to be dependent on the major androgen receptor (AR); similar to Ferguson’s group, Loeuillet et al. found no effect from manipulating estrogen receptors. It is somewhat curious that the scrapie effect seemed to involve some interaction in the periphery (peripheral interactions of PrP with the product of an AR-induced gene would be a reasonable hypothesis, given the effects of both AR mutation and castration); latency after direct CNS injections was not sex-dependent. Effects on mGluR5 were observed in dissociated cell cultures, again suggesting a distinction between scrapie propagation and this neurotransmission hypothesis. At any rate, manipulation of AR in this AD model would be intriguing.
References:
Loeuillet C, Boelle PY, Lemaire-Vieille C, Baldazza M, Naquet P, Chambon P, Cesbron-Delauw MF, Valleron AJ, Gagnon J, Cesbron JY. Sex effect in mouse and human prion disease. J Infect Dis. 2010 Aug 15;202(4):648-54. PubMed.
Make a Comment
To make a comment you must login or register.