Suspected Non-Alzheimer Pathophysiology: It’s Not Exactly a Snap
Quick Links
Suspected non-Alzheimer's pathophysiology has puzzled the field since researchers coined the term three years ago. SNAP describes cognitively normal older adults who have one of several markers of neurodegeneration but test negative for brain amyloid and have not been diagnosed with a specific neurodegenerative disorder (Jack et al., 2012). While it seems clear that SNAP is not a preclinical stage of AD, scientists are having a hard time deciding what it is. At this year's Alzheimer’s Association International Conference, held July in Washington, D.C, SNAP was a focus of presentation and debate. Some researchers reported that SNAP might correlate with other morbidities such as cerebrovascular disease. Others believe SNAP progresses very slowly or not at all, and in some cases might reflect normal aging. Most agreed that the definition of aging versus neurodegeneration might have to change, because the biomarkers used to measure degeneration each reflect slightly different processes. "While we know that some degree of brain atrophy occurs with age, we don’t really know what aging is and we really need to understand that better," said Bill Jagust, University of California, Berkeley.
SNAP has been documented in several large studies, including the Mayo Clinic Study of Aging, the Berkeley Aging Cohort, the Alzheimer's Disease Neuroimaging Initiative, the Alzheimer's Disease Research Center at Washington University, St. Louis, and others. Consistently, up to a quarter of cognitively normal older adults in these cohorts show signs of neurodegeneration on FDG PET scans, on structural MRIs of the hippocampus, or by CSF tau, while falling below the threshold for a positive amyloid scan in the brain. In this way, SNAP posed a challenge to the National Institute on Aging–Alzheimer's Association (NIA-AA) proposed diagnostic criteria for preclinical AD. They classify people into sequential stages of having: no abnormality (stage 0); amyloid (stage 1); amyloid and evidence of neurodegeneration (stage 2); or amyloid, neurodegeneration, plus subtle cognitive/behavioral decline (stage 3, Sperling et al., 2011). What causes SNAP, and what does it mean for people who have it?

SNAP Is Not Progressive.
As measured by the cognitive battery of AIBL, people with SNAP (blue) start out slightly worse than controls (green), but stay stable over the course of six years. The yellow and red groups represent preclinical Alzheimer’s. [Courtesy of Samantha Burnham, University of Melbourne, Australia.]
Researchers are beginning to refine the clinical picture of SNAP and follow it longitudinally. At AAIC, Beth Mormino, Massachusetts General Hospital, Charlestown, reported on SNAP in the Harvard Aging Brain Study. As with other cohorts, a quarter of the 242 people enrolled fell into this category. At an average age of 76, they tended to be older than cognitive normals who tested negative for neurodegeneration, and less likely to carry an ApoE4 risk allele than normals who were positive for amyloid. This was true in other cohorts, as well. Mormino tested for several morbidities that might explain the neurodegeneration markers in the SNAP group, but found no correlation with cardiovascular disease risk factors such as high body-mass index or white-matter hyperintensities (WMH) in the brain. Likewise there was no correlation to tau tangles in various regions of the brain, including in the medial-temporal lobe, the entorhinal cortex, and parahippocampal gyrus. Mormino found that people with SNAP and age-matched controls who test negative for both amyloid and markers of neurodegeneration (i.e., stage 0 for AD) similarly take up the neurofibrillary tangle tracer T-807 in the brain. "At that age, tangles in the medial temporal lobe of the brain are prevalent, even in people at stage 0," Mormino said. "While there could be tau pathology in SNAP, it does not seem specific to it," she added.
The findings suggest that SNAP is not the same as PART, aka primary age-related tauopathy, which was evoked to describe tangle pathology in the absence of Aβ pathology (see Nov 2014 news).
Melissa Murray, Mayo Clinic, Jacksonville, Florida, agreed that tau pathology does not fully explain SNAP. Murray autopsied 65 people who had taken part in the Mayo Clinic Study of Aging or enrolled in the Mayo ADRC. During life, 11 had been diagnosed with SNAP based on glucose hypometabolism in FDG PET scans. At death, none of them fulfilled NIA-AA pathological criteria for Alzheimer's disease, supporting the SNAP designation. What explains the neurodegeneration marker? Postmortem pathology revealed that one person had had hippocampal sclerosis, four argyrophilic grain disease, and six fulfilled criteria for PART.
Murray’s data supports the idea that tau pathology alone cannot explain all SNAP cases. In fact, seven of these 11 had moderate to severe arteriosclerosis and white-matter disease, she said, though it is not clear how these pathological findings relate to SNAP. The timing of the autopsies complicates analysis of this data. The volunteers were 83 years old, on average, when they had their FDG PET scan, and 87 when they died. Of the 11 cases, five came to autopsy within three years of the initial SNAP diagnosis, but the delay was longer for the other six so some of the pathology could have developed after the scan.
Previous analysis of the Mayo cohort had reported that patterns of WMHIs in SNAP tracked more closely with those in stage 2/3 preclinical AD than those in stage 0/1. The incidence of hypertension and diabetes were also similar in SNAP and stage 2/3 AD, and higher than in the stage 0/1 (see Knopman et al., 2012). These findings indicate that neurodegeneration in cognitively normal individuals may be related to vascular pathology, said Miranka Wirth, Inserm, Caen, France, who is now at Charité University Medicine in Berlin.
To explore this relationship, Wirth compared hippocampal volume, glucose metabolism, and regional cortical atrophy with white-matter lesions in people enrolled in the Berkeley Aging Cohort. In people without brain amyloid, the size of the white-matter lesions correlated with thinning of the cortex; intriguingly, however, their cortex thinned out in regions that do not typically atrophy in AD. Conversely, in people with brain amyloid, white-matter lesions exacerbated cortical thinning in those areas of the brain that typically accumulate Aβ. This is a finding Sylvia Villeneuve had previously reported while working with Wirth at Bill Jagust's lab (Villeneuve et al., 2014).
Cerebrovascular disease also appears more common in SNAP cases than in controls in the Australian Imaging, Biomarkers, and Lifestyle (AIBL) study. In Washington, Samantha Burnham from the Commonwealth Scientific and Industrial Research Organisation (CSIRO) in Melbourne, Australia, reported among that among 132 AIBL participants diagnosed with SNAP, 36 percent tested positive for WMHIs, a much higher proportion than in normal elderly controls.
Taken together, the SNAP versus AD patterns of imaging and fluid markers suggest that vascular factors can accelerate neurodegeneration in vulnerable brain regions, which vary depending on the presence of a disease-specific pathology such as Aβ.
Does SNAP Get Worse?
Based on current data, it appears that cognition in people with SNAP deteriorates much more slowly, if at all, than in those who have both brain amyloid and neurodegeneration. In keeping with this idea, Mormino reported at AAIC that the practice effect in the Harvard Aging Brain Study, aka the ability of cognitively normal people to score better on repeat testing, diminishes in those with SNAP, much like it does in people at stage 1 AD. At a group level, about 15 percent of people with SNAP declined to CDR 0.5 over an average of two years. Mormino said that is about the same rate as people with stage 1 AD, but much slower than people with stage 2. Researchers at the Mayo Clinic and at Washington University have seen similar rates of decline, she said. More recently, Mormino measured change in specific cognitive domains. Executive function and memory declined in SNAP at about the same rate as in people with stage 1 AD. "The pattern is always the same," said Mormino.
In the AIBL study, people with SNAP decline even more slowly, Burnham reported. She followed 545 cognitively normal healthy controls, testing them with the Preclinical Alzheimer's Cognitive Composite, a battery that measures executive function, episodic memory, and global cognition (see June 2014 news). The 132 people designated as SNAP had slightly poorer scores at baseline, but maintained their cognition over the next seven years, just like controls who tested negative for both amyloid and markers of neurodegeneration (see image above). "SNAPs do not decline and behave over time like the Aβ-negative/neurodegeneration-negative group," said Burnham.
If not cognition, does neurodegeneration worsen in SNAP? Not much, apparently. At an AAIC imaging preconference, Pierrick Bourgeat from CSIRO, Brisbane, Australia, reported atrophy rates among 320 people enrolled in AIBL. This dataset has a full eight years of follow-up. Hippocampal volume fell fastest in 11 people who tested positive for both amyloid and neurodegeneration (stage 2). In 48 people who had evidence of amyloid but none of neurodegeneration, the hippocampus shrank more slowly. In contrast, in the 55 SNAP cases, hippocampal volume behaved quite differently. It started out low, but did not shrink over the course of eight years.
Bourgeat reported the same pattern for temporal and parietal cortex atrophy, with fast decline in the group with both pathology markers and slowest in SNAP. Other cortical regions trended the same way. Likewise, ventricular volumes expand fastest in people with amyloid/neurodegeneration markers and slowest in SNAP.
The upshot of all this, said Bourgeat, is that SNAP follows a wholly different trajectory to Alzheimer's disease (see image below).
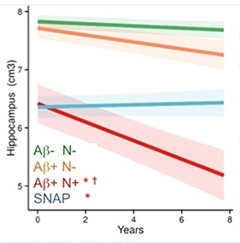
More than a Snapshot.
Over eight years, there was no atrophy in people with SNAP. [Courtesy of Pierrick Bourgeat, University of Melbourne, Australia.]
Then what in the world is SNAP? In his talk, Jagust cautioned that the construct is artificial. He noted that scientists apply a bimodal distribution for amyloid, classifying it as either positive or negative based on a specific cut-off, but treat hippocampal volume and glucose metabolism as continuous measures. "It is clear to me that SNAP is a biomarker construct that classifies people based on thresholds for Aβ and biomarkers that have no natural cut point," Jagust said. "A lot may depend on thresholds that are arbitrarily chosen."
On top of that, biomarkers of neurodegeneration, be they hypometabolism, atrophy, or neuronal damage reflected as CSF tau, are not necessarily equivalent. They all measure slightly different things and only modestly correlate with each another, noted David Knopman, Mayo Clinic, Rochester, Minnesota. "The field could get itself into trouble if investigators are not appropriately cautious about specifying which neurodegeneration biomarker they are referring to." In fact, Brian Gordon and colleagues at WashU reported that among 212 normal older adults (mean age 67), CSF tau and hippocampal volume correlated poorly, agreeing only 57 percent of the time. "Different people have either one or the other marker. They are not equivalent," said Gordon. Because high CSF tau and low hippocampal volume are not concordant, i.e., they don't always occur in the same person, it is not clear that they reflect AD pathology, he said. By comparison, amyloid PET and CSF Aβ42 are highly concordant, Gordon said.
This raises the question of exactly what causes the positive neurodegeneration marker in SNAP to begin with. "A lot of different biological processes can lead to abnormal hippocampal volume or abnormal CSF tau," said Gordon. "Unlike amyloid, those processes are not AD specific." The markers might reflect underlying cardiovascular disease, argyrophilic grain disease, hippocampal sclerosis, diabetes, stress, or some yet-to-be-discovered problem. Jagust noted that a certain amount of atrophy occurs with age (see Jun 2013 news).
In short, scientists don't know what causes the neurodegenerative signal in SNAP. Victor Villemagne, University of Melbourne, believes it may be unrelated to AD. "For me, the SNAP findings demonstrate that there is no 'no-Aβ' pathway to AD. You might have some degree of cognitive impairment and 'AD-like' neurodegeneration, but if Aβ is not present you have a completely different progression over time compared to when it is present, suggesting two completely different underlying pathophysiologies." With SNAP, we can now identify people who definitely do not belong in AD therapeutic trials, he added. Vice versa, many scientists agreed that AD therapy trials may want to refrain from using structural MRI as an inclusion criterion. If an AD trial enrolls patients due to their low baseline hippocampal volume, then it may end up with SNAP participants who do not have AD and will barely progress during the duration of the trial.
If SNAP does not progress over the course of eight years, can it be called neurodegeneration? "There is some reason for the abnormalities, but it is not progressive neurodegeneration," said Gordon. Jagust agreed that having a small hippocampus may be unrelated to a progressive disease and recommended that SNAP be reconsidered. Jagust suggested incorporating evidence of neurodegeneration into SNAP. Categorizing subjects by longitudinal change would undoubtedly alter the proportion of people who are called SNAP and may be a better, and more specific, index of brain degeneration, Jagust said.
"We do know that if you have neurodegeneration plus Aβ in the brain, then you are in trouble," said Jagust. "The key question is can you get neurodegeneration without Aβ? That's the fundamental question. We need to follow people longitudinally to answer it."—Tom Fagan
References
News Citations
- Scientists Propose a New Definition for Tau-Only Pathology
- Test Battery Picks Up Cognitive Decline in Normal Populations
- Brain Changes Speak Volumes About Normal Aging and Dementia
Paper Citations
- Jack CR, Knopman DS, Weigand SD, Wiste HJ, Vemuri P, Lowe V, Kantarci K, Gunter JL, Senjem ML, Ivnik RJ, Roberts RO, Rocca WA, Boeve BF, Petersen RC. An operational approach to National Institute on Aging-Alzheimer's Association criteria for preclinical Alzheimer disease. Ann Neurol. 2012 Jun;71(6):765-75. PubMed.
- Sperling RA, Aisen PS, Beckett LA, Bennett DA, Craft S, Fagan AM, Iwatsubo T, Jack CR Jr, Kaye J, Montine TJ, Park DC, Reiman EM, Rowe CC, Siemers E, Stern Y, Yaffe K, Carrillo MC, Thies B, Morrison-Bogorad M, Wagster MV, Phelps CH. Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011 May;7(3):280-92. Epub 2011 Apr 21 PubMed.
- Knopman DS, Jack CR, Wiste HJ, Weigand SD, Vemuri P, Lowe VJ, Kantarci K, Gunter JL, Senjem ML, Mielke MM, Roberts RO, Boeve BF, Petersen RC. Brain injury biomarkers are not dependent on β-amyloid in normal elderly. Ann Neurol. 2012 Nov 23; PubMed.
- Villeneuve S, Reed BR, Madison CM, Wirth M, Marchant NL, Kriger S, Mack WJ, Sanossian N, DeCarli C, Chui HC, Weiner MW, Jagust WJ. Vascular risk and Aβ interact to reduce cortical thickness in AD vulnerable brain regions. Neurology. 2014 Jul 1;83(1):40-7. Epub 2014 Jun 6 PubMed.
Further Reading
Annotate
To make an annotation you must Login or Register.

Comments
No Available Comments
Make a Comment
To make a comment you must login or register.