Mini Strokes Cause Mega Problems for Brain Cleansing
Quick Links
Microinfarcts may be puny, but they pack a punch when it comes to brain cleaning, according to new research. In the March 15 Journal of Neuroscience, scientists led by Maiken Nedergaard, University of Rochester Medical Center, New York, and Jeffrey Iliff, Oregon Health & Science University, Portland, report that in mice, teeny strokes have a broad impact on cleansing throughout the brain. Such microinfarcts also occur in people but normally go unnoticed. Even infarcts restricted to one hemisphere impaired glymphatic clearance on the contralateral side. What’s more, solutes that collected in and around these microinfarcts might accelerate protein aggregation in Alzheimer’s and other forms of neurodegeneratn, the researchers claim. In keeping with this idea, the effect of microinfarcts on clearance worsened as mice aged. The results offer one reason why so-called “silent strokes” are associated with risk for Alzheimer’s.
“This is just one more piece of evidence that microinfarcts actually contribute to dementia, and that their effects on surrounding tissues are much broader than we think,” said Andy Shih, Medical University of South Carolina, Charleston.

Making Microinfarcts.
Three days after cholesterol crystals are injected into the carotid artery, microinfarcts (arrows) appear in the mouse brain, spurring local activation of microglia and macrophages (red) and more diffuse markers of astrogliosis (blue). [Reprinted with permission: Wang, et al. The Journal of Neuroscience 2017.]
Various types of brain injury, traumatic or from hemorrhaging or large strokes, impair glymphatic clearance (Gaberel et al., 2014; Iliff et al., 2014). This regular flushing of the brain, proposed to clear out extracellular waste such as Aβ, occurs when cerebrospinal fluid flows adjacent to arteries, diffuses into the brain parenchyma, and rushes out toward veins, carrying cell waste that had been lingering in the interstitial fluid (Aug 2012 news). This movement requires the aquaporin-4 water channel on the end feet of astrocytes that line blood vessels. However, this group previously reported that microinfarcts suppress the water channel there (Wang et al., 2012). Could tiny areas of dead tissue also impair glymphatic flux?
To find out, first author Minghuan Wang and colleagues induced microinfarcts in two- to three-month-old wild-type mice by injecting cholesterol crystals, 40 to 70 μm in size, into the right carotid artery. This debris lodged in tiny vessels on the same side of the brain, mostly in the deep cortex, hippocampus, and subcortical structures, cutting off circulation in the immediate vicinity. After three days, these microinfarcts had a core of microglia and macrophages and were surrounded by widespread activated astroglia (see image above). Control mice injected with saline appeared normal.
To visualize the effects on the glymphatic system, the researchers injected a fluorescent tracer into the CSF, then monitored clearance in two ways. For surface clearance, they watched through a glass-covered window in the mouse brain using two-photon imaging. For a deeper view, they sacrificed the animals just after injecting the CSF tracer, then looked at brain slices with fluorescence microscopy. Both assays revealed that CSF flowed freely in controls on both sides of the brain. However, it all but halted in the ipsilateral hemisphere of animals that got the crystals and slowed considerably on the contralateral side, too (see image below).
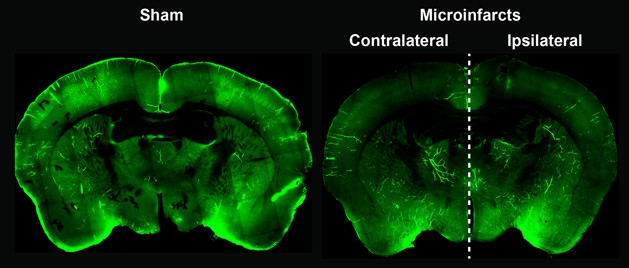
Impaired Clearance. Inducing microinfarcts in the mouse brain weakens glymphatic function throughout the brain (green) compared to controls. [Reprinted with permission: Wang, et al. The Journal of Neuroscience 2017.]
Would aging exacerbate the effect of microinfarcts on the glymphatic system? This group previously reported that aging itself takes a toll on glymphatic flow in mice (Kress et al., 2014). To test this question, Wang and colleagues repeated their cholesterol crystal experiment in 12-month-old mice. By this age, clearance in controls had slowed to a pace comparable to the crystal-injected young adults. With the addition of microinfarcts, glymphatic function dropped even further, especially in the hippocampus and cortex.
While the effects were far-reaching, they were temporary. Within two weeks, flow returned to normal in both young and old treated animals. However, the microinfarcts had begun to accumulate solutes, which remained at the two-week mark. This hints that molecules such as Aβ and tau may pool around infarcts in the same way, and could help explain why microinfarcts heighten risk for Alzheimer’s, said Nedergaard. “If you are older, and on top of that you have microinfarcts, you’re probably clearing even fewer of these proteins,” she told Alzforum. Shih was intrigued. “This may be one persistent effect of these lesions, that they harbor some damaging molecules and cause long-term inflammation.”
Nedergaard was surprised that the effects were so extensive across the brain. She theorized that even distant astrocytes could become activated, losing end-feet AQP4 channels. In fact, last year Iliff’s group reported that a dearth of AQP4 from astrocyte end feet correlated with cognitive decline in people with Alzheimer’s disease (see Dec 2016 news). Shih proposed an alternate mechanism, suggesting that some of the cholesterol crystals themselves crossed to other side of brain and created subtle problems that affected glymphatic function. He wondered if scientists would see the same global effects with a more targeted model, perhaps one that uses lasers to induce microinfarcts in specific spots (Summers et al., 2017).
Nedergaard said her group will next test mouse models with other types of underlying vascular disease. Costantino Iadecola, Weill Cornell Medical College, New York, said it would be interesting to do the experiment in an APP mouse to see if microinfarcts exacerbate accumulation of Aβ. “This [work] raises the possibility that failure of protein clearance may contribute to the cognitive dysfunction caused by microinfarcts,” he told Alzforum. It would fit with the idea that clearance of Aβ is impaired in sporadic AD, he said. It would also help explain why aging and diffuse ischemic injury may bring on the disease, he added. To probe the potential mechanism, he suggested testing whether glymphatic suppression correlated with gliosis and loss of APQ4.
Other researchers are working on ways to visualize glymphatic clearance in people (Eide and Ringstad, 2015). Studying flow in people who develop microinfarcts, reduce their blood pressure, or have AD would enhance our understanding of the effects on the glymphatic system in disease, Nedergaard said.

Migraine Impairs Glymph. In a mouse model of migraine (bottom), a fluorescent dextran (green) injected into the cortex flows poorly into the paravascular space around the pial artery. In control mice (top), it fills the space within a few hours. [Reprinted with permission: Schain, et al. The Journal of Neuroscience 2017.]
A second paper in the same issue of the Journal of Neuroscience suggests that glymphatic clearance comes into play in migraine headaches, which have been linked to cognitive decline (see Rist et al., 2013, for a review). Just before a migraine, a slow wave of abnormal neural activity propagates across the brain. Researchers led by Rami Burstein at Beth Israel Deaconess Medical Center, Boston, induced this wave, called cortical spreading depression, in mice via a pinprick to an exposed area of the cortex, then looked at their blood vessels using in vivo two photon imaging. First author Aaron Schain and colleagues saw that the paravascular space immediately surrounding the brain’s blood vessels, through which CSF flows, closed up completely, halting glymphatic flow for several minutes. It gradually recovered over the next half hour. The results suggest a link between the glymphatic system and migraine, and hint at one way that the brain regulates glymphatic flow.—Gwyneth Dickey Zakaib
References
News Citations
- Brain Drain—“Glymphatic” Pathway Clears Aβ, Requires Water Channel
- Dearth of Water Channels a Sign of ‘Glymphatic’ Breakdown in Alzheimer’s?
Paper Citations
- Gaberel T, Gakuba C, Goulay R, Martinez De Lizarrondo S, Hanouz JL, Emery E, Touze E, Vivien D, Gauberti M. Impaired glymphatic perfusion after strokes revealed by contrast-enhanced MRI: a new target for fibrinolysis?. Stroke. 2014 Oct;45(10):3092-6. Epub 2014 Sep 4 PubMed.
- Iliff JJ, Chen MJ, Plog BA, Zeppenfeld DM, Soltero M, Yang L, Singh I, Deane R, Nedergaard M. Impairment of glymphatic pathway function promotes tau pathology after traumatic brain injury. J Neurosci. 2014 Dec 3;34(49):16180-93. PubMed.
- Wang M, Iliff JJ, Liao Y, Chen MJ, Shinseki MS, Venkataraman A, Cheung J, Wang W, Nedergaard M. Cognitive deficits and delayed neuronal loss in a mouse model of multiple microinfarcts. J Neurosci. 2012 Dec 12;32(50):17948-60. PubMed.
- Kress BT, Iliff JJ, Xia M, Wang M, Wei HS, Zeppenfeld D, Xie L, Kang H, Xu Q, Liew JA, Plog BA, Ding F, Deane R, Nedergaard M. Impairment of paravascular clearance pathways in the aging brain. Ann Neurol. 2014 Dec;76(6):845-61. Epub 2014 Sep 26 PubMed.
- Summers PM, Hartmann DA, Hui ES, Nie X, Deardorff RL, McKinnon ET, Helpern JA, Jensen JH, Shih AY. Functional deficits induced by cortical microinfarcts. J Cereb Blood Flow Metab. 2017 Jan 1;:271678X16685573. PubMed.
- Eide PK, Ringstad G. MRI with intrathecal MRI gadolinium contrast medium administration: a possible method to assess glymphatic function in human brain. Acta Radiol Open. 2015 Nov;4(11):2058460115609635. Epub 2015 Nov 17 PubMed.
- Rist PM, Kurth T. Migraine and cognitive decline: a topical review. Headache. 2013 Apr;53(4):589-98. Epub 2013 Feb 13 PubMed.
Further Reading
Primary Papers
- Wang M, Ding F, Deng S, Guo X, Wang W, Iliff JJ, Nedergaard M. Focal Solute Trapping and Global Glymphatic Pathway Impairment in a Murine Model of Multiple Microinfarcts. J Neurosci. 2017 Mar 15;37(11):2870-2877. Epub 2017 Feb 10 PubMed.
Annotate
To make an annotation you must Login or Register.

Comments
University of Southampton School of Medicine
University of Southampton School of Medicine
In this study, Wang et al. created microinfarcts in the brain at three days after injection of cholesterol crystals into the carotid artery. Infarcts were identified by the presence of macrophages and reactive astrocytosis.
Following injection of tracer into the CSF of the cisterna magna, influx of tracer alongside arteries into the surface of the brain was significantly reduced at 30 minutes after injection in the animals with multiple infarcts. Such impairment was greater in animals at 12 months of age compared with those at two to three months of age. Tracer accumulated within infarcts, presumably within macrophages, although this is not stated. No experiments examined the pathways of removal of tracer from the brain.
This is a valuable new model of microinfarction, a common feature of small vessel disease, comparable to the models produced in recent years by the groups led by Murphy and Ihara (Silasi et al., 2015; Okamoto et al., 2012). Assessment of glymphatic/convective influx of the fluorescent tracer after 30 minutes of its injection into the CSF compartment showed the tracer in association with the perivascular compartment of arteries, suggesting that solutes that enter the brain from the CSF along the pial-glial basement membranes, are then directed toward the intramural periarterial drainage pathways. Cholesterol crystals occluding cerebral arteries as well as the age-related arterial stiffness appear to interfere with the motive force for both glymphatic entry of CSF and intramural periarterial drainage.
The discussion is rather speculative, and citation of the literature concerned with entry and elimination of fluid and solutes into and from the brain is selective and restricted. It is not clear how the results of this paper can be translated into the development of treatments for multi-infarct dementia or other dementias.
References:
Silasi G, She J, Boyd JD, Xue S, Murphy TH. A mouse model of small-vessel disease that produces brain-wide-identified microocclusions and regionally selective neuronal injury. J Cereb Blood Flow Metab. 2015 May;35(5):734-8. Epub 2015 Feb 18 PubMed.
Okamoto Y, Yamamoto T, Kalaria RN, Senzaki H, Maki T, Hase Y, Kitamura A, Washida K, Yamada M, Ito H, Tomimoto H, Takahashi R, Ihara M. Cerebral hypoperfusion accelerates cerebral amyloid angiopathy and promotes cortical microinfarcts. Acta Neuropathol. 2012 Mar;123(3):381-94. Epub 2011 Dec 15 PubMed.
Make a Comment
To make a comment you must login or register.